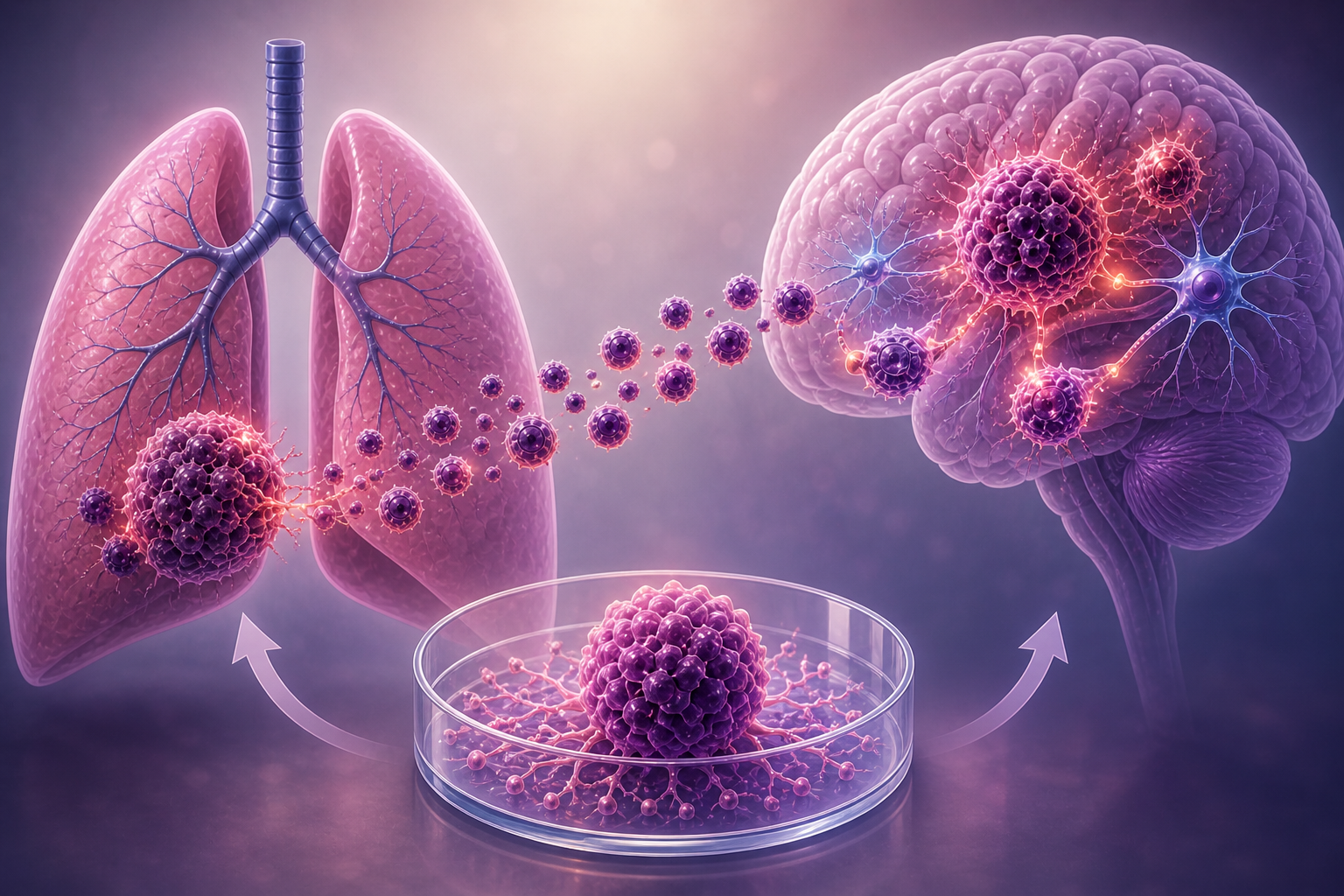
Lung Cancer — those two words hold a lot of weight. Two words each person hopes to never hear, at least not in association with themselves, or anyone they love. Yet, these words are a diagnosis for about 2.5 million people worldwide [1]. For the roughly 350,000 people who get diagnosed with small-cell lung cancer (SCLC), this is a death sentence [1].
Despite the name, this cancer is not limited to the lungs. With striking progression, SCLC can travel throughout the body, with a notable preference for the brain. The survival rate of this cancer is grim; however, recent scientific advancements may provide a light at the end of the tunnel.
How is SCLC Different From Other Forms of Lung Cancer?
There are two types of lung cancers, differentiated by the size of cells that make up the associated tumors. The first is small-cell lung cancer (SCLC), which makes up approximately 15% of all lung cancer. The other is non-small-cell lung cancer (NSCLC), consisting of the remaining 85% of lung cancer diagnoses [1, 2]. Patients with SCLC may experience respiratory symptoms such as a cough or shortness of breath. In some cases, patients will experience neurological symptoms, such as seizures, headaches, cognitive function changes, motor deficits, or personality changes [2]. However, approximately 60% of patients with SCLC exhibit virtually no symptoms at diagnosis, making the disease difficult to treat in the time it would be the easiest to intervene [2, 3]. Most SCLC patients will not live more than 5 years after they are diagnosed with the disease [4].
SCLC is strongly linked to frequent tobacco exposure, with approximately 95% of SCLC occurring in people with a history of smoking [5]. This is because cigarette smoke contains chemicals that can damage DNA, causing the DNA of heavy smokers to become fragile and susceptible to continuous gene mutations [6, 7]. Genes provide instructions for creating all biological elements in the human body. Therefore, altering these instructions will change how the body is supposed to function, which can have catastrophic effects.
Catastrophic Cancerous Cell Cycle
In a healthy human body, cells have several mechanisms that tell them when to stop multiplying. The genes that encode the critical proteins that regulate these mechanisms are called tumor suppressor genes [6]. When there is a mutation in a tumor suppressor gene, this process is no longer regulated, and cells will continue to divide and multiply. Once there is no longer space for the cells to line up next to each other, they will begin to stack on top of each other. These cells are building a tumor [6, 7].
SCLC has a higher rate of gene mutations than any other form of cancer. In SCLC, this process is rapid due to the high number of mutations that alter DNA’s instructions [7]. Cancerous cells can grow and multiply at an extremely fast rate, leading to lots of different variations of cells with unique instructions encoded in their genes. Some of these cells will be specialized in specific functions, whereas others will have more generalizable functions, meaning they are more adaptable, but less efficient [8]. These generalist cells are more apt to change identity and survive under stress.
SCLC may initially respond well to treatments, such as chemotherapies and immunotherapies, which aim to kill the cancerous cells and prevent them from multiplying [4, 9]. However, since SCLC cells replicate and change so rapidly, generalist cells can adapt to withstand the therapies, causing the cancer to become resistant to the treatment [9, 10]. This leads to an awful cycle of tumor growth, spread and resistance.
There is Nothing Small About SCLC.
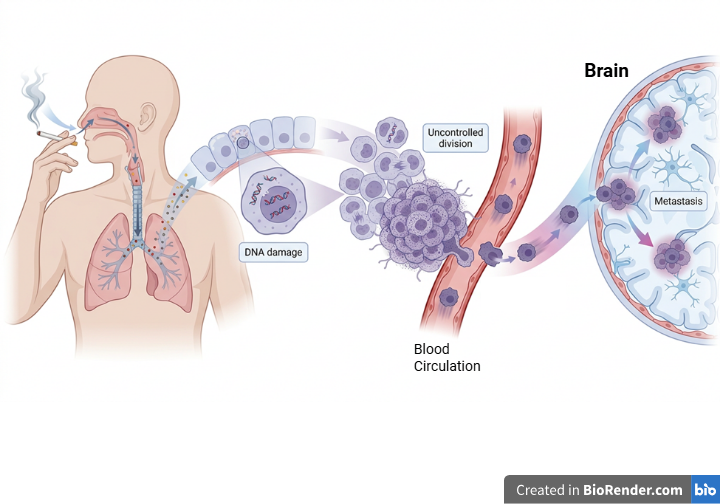
As the disease progresses, tumor cells can separate from the lungs and invade local tissue. The cancer cells are quick to take advantage of the lung’s large blood supply [11]. In a process known as metastasis, the cancer cells hitch a ride to other parts of the body through the bloodstream, where they will colonize new environments [12]. Often, this process will have already begun before a patient even knows that he or she has the disease [9].
One of the environments that SCLC strongly favors is the brain. At diagnosis, approximately 15% of SCLC cancer has already traveled to the patients’ brains [1, 7]. Two years after diagnosis, this estimate will have increased to as many as 80% of patients [11].
Created in FigureLabs and https://BioRender.com
A critical component of this process is the blood-brain barrier. This barrier is a tight structure that separates the brain from the body’s main blood stream [13]. SCLC tumor cells can penetrate this barrier and integrate themselves into the brain’s natural circuitry [8].
Many SCLC cells do not survive for long after crossing the blood–brain barrier, however, those that do are able to quickly adapt [8, 11]. In the brain, these adapted cells encounter an environment that provides nutrients that seem to perfectly support their survival. These invading cells quickly take up residence, consuming critical resources within the brain [9].
Part of the reason SCLC cells favor the brain is because they are neuroendocrine in nature, meaning they possess properties of both nerve cells and hormone‑producing cells [3, 7]. These properties provide the cancerous cells with an effective disguise, taking on characteristics that are more like brain or nerve cells than lung cells [14].
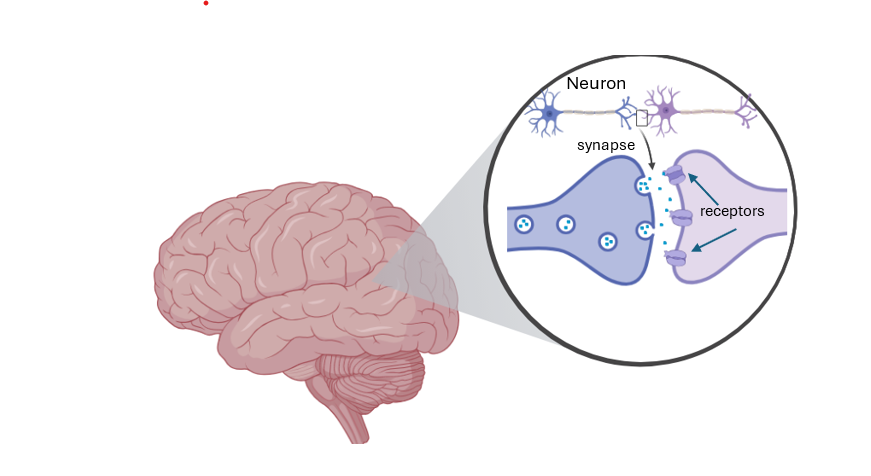
The brain sends and receives chemical messages through nerve cells, called neurons. These neurons connect with one another through synapses, where chemical messengers are released and bind to specific proteins on the surface of neighboring cells. These proteins, known as receptors, allow neurons to detect and respond to signals.
| Created in FigureLabs and https://BioRender.com |
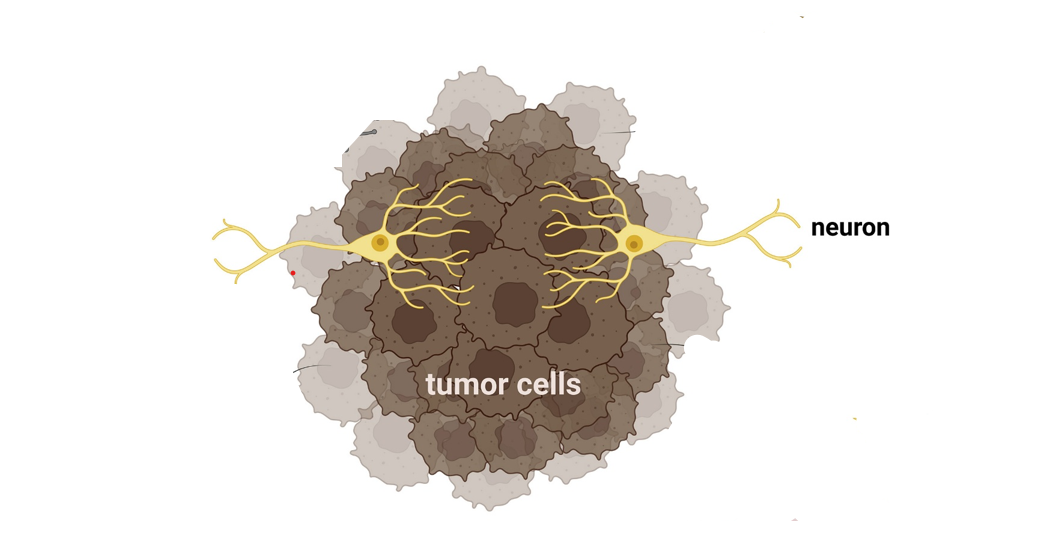
Some SCLC cells are able to hijack the brain’s natural processes. These cancerous cells will form connections with neurons that release a specific chemical messenger known as Glutamate [2]. Like undercover spies, these cancerous cells will produce proteins that mimic Glutamate receptors, allowing the growing tumor to intercept the chemical messages. Unknowingly, the brain’s neurons will form a synapse with the cancer cells, releasing Glutamate to bind to receptors on the tumor cells’ surface [2].
This is all a ploy, carefully implemented by the cancerous cells. When the SCLC cells receive a lot of Glutamate signals, changes can occur in the surrounding tumor environment that support cancer survival and continual tumor growth [2, 3]. This enhanced Glutamatergic signal can also cause neurological symptoms that are often seen in patients with SCLC brain metastasis, such as seizures [2].
This hijacking of glutamate signaling also consumes huge amounts of energy, which can be depleted from other sources of the brain that are critical for survival. With this huge energy consumption and redistribution of resources, the brain is no longer able to function normally, leading to a rapid decline in patient health. Most people will not survive more than 5 months after brain metastases have been identified [14].
Unfortunately, there are many barriers to out-maneuvering these sleuthing cells. Since patients’ life expectancies are so limited by time the brain metastases are identified, there are not many candidates for surgical removal, making it difficult to uncover new ways to treat the disease [9, 10]. At that point in time, medical intervention is primarily focused on providing comfort and preserving quality of life.
Additionally, these cancerous cells rarely survive outside of the brain [10, 15]. If scientists manage to capture the cells and sustain their survival outside of the human body, the cells are generally very finicky and prone to change, often shifting to act differently without the support of the brain’s communication networks. [8, 15]. This requires researchers to take new approaches to curing SCLC at different stages of discovery.
But wait! There is Hope!
Believe it or not, this article is not being written just to tell you just how horrible this cancer is, and how hopeless we are at eradicating it. We all know cancer is terrible, but what are we doing about it? The answer is a lot. In fact, recent years have led to several promising advancements towards combating this terrible disease.
One of these advancements is something called organoid models. These models are basically 3-dimensional miniature models of a patient’s tumor, directly created from the patient’s tumor tissue [8].
Much like how seeds are extracted from a grown plant, provided water, nutrients, and sunlight to produce a new plant, these organoid models are created by extracting cells from a patient’s tumor [8, 15]. These cell seedlings are then nurtured in a dish and given specific nutrients that help to mimic the conditions of the human body. These nutrients help the tumor cells multiply and grow into an organoid, which resembles the original tumor.
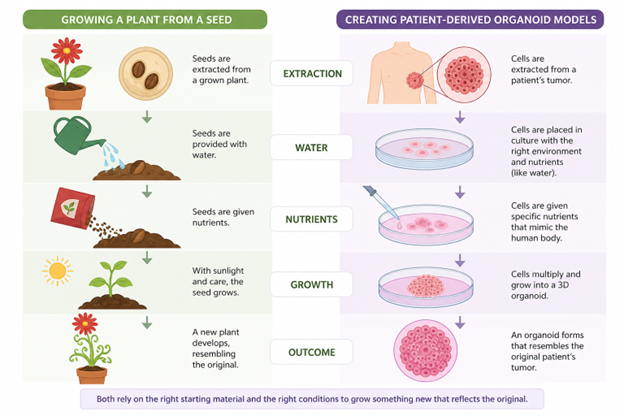
Created by FigureLabs
By creating these organoids, researchers can investigate the most effective methods of combatting SCLC, as if these treatments are tested directly on the patients. These models allow scientists to determine whether patients respond better to some treatments than others, or whether specific interventions are necessary [8]. Ultimately, these tumor clones offer an avenue of personalized medical care that better supports patients’ individual differences.
Modeling SCLC Brain Metastasis
Rather than replicating the SCLC brain metastasized tumors, scientists have now taken the route of creating the entire brain from scratch and implanting the cancer into this freshly created brain organoid [12, 15]. Scientists can use human embryonic stem cells, which come from very early human embryos, to create a miniature 3-dimensional brain. Since these cells are in their very early stages of life, they are capable of evolving into virtually any type of cell in the body [16]. With the correct signals and environment, scientists can artificially induce these changes so that the embryonic cells will develop into brain cells. Those cells will eventually multiply and naturally organize themselves into a miniature 3-dimensional brain.
Once scientists create the environment of the brain, they can introduce cancerous cells into the synthetic brain. To ensure that the progression of SCLC is trackable, scientists will specifically engineer the cancer cells to light up with a red fluorescence [8, 12]. This 3-dimensional model allows researchers to directly examine interactions between SCLC cells and neurons that drive the disease progression.
These brain organoids closely resemble human brain development over time, and patient-specific cancer, so that when tumors are grown inside the organoids, they behave much like metastasized SCLC that preserves patient biology [17]. Researchers have demonstrated that different variations of SCLC cells can rapidly invade the mini brain models and spread throughout the tissue in just days [8]. This model can preserve the diversity and adaptability of the disease seen in real patients, providing new avenues for scientific advancement [8].
The Light at the End of the Tunnel
Organoid models have opened the door for rapid advancements in SCLC treatments. Recent studies have demonstrated that these individualized patient tumor organoid models can accurately predict patent-specific responses to chemotherapy and targeted therapies [17]. For example, scientists can now link certain patient-specific gene mutations to therapeutic efficiency [18]. These predictive abilities suggest these models can act as a powerful tool for personalized care.
This advanced modeling can also allow researchers to validate the mechanisms involved SCLC and brain metastasis, to identify actionable targets for intervention [8]. These models can also guide scientific understanding of SCLC adaptability, tumor progression, and patient-specific treatment responses, positioning these models as avenues for new discoveries [8, 17].
Among these discoveries, a recent study found that blocking the activation of SCLC Glutamate receptors can slow the progression of SCLC brain metastasis [2]. Additionally, SCLC brain organoid models have led researchers to identify signals that can act as critical components of cancerous cell survival and progression. Specifically, SCLC seems able to recruit specific cells inside the brain that release signals promoting cancer growth and survival [9,10]. Scientists are now working to develop therapies that target those cells to make the conditions of the brain much less “livable” for SCLC cells.
Small-cell lung cancer is no small feat. For much of its history, this disease has been treated as a death sentence, with limited therapeutic options and poor patient outcomes. However, new hope is emerging as science continues to advance and innovate new solutions that tackle this massive problem from new angles. What once seemed like science fiction, such as tumor cloning and lab-grown brains, are now a reality. These technologies stand at the forefront of modern cancer research, offering a deeper understanding of SCLC biology while revealing actionable therapeutic targets and advancing personalized patient care.
References
[1] Bray, F., Laversanne, M., Sung, H., Ferlay, J., Siegel, R. L., Soerjomataram, I., & Jemal, A. (2024). Global cancer statistics 2022: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 74(3), 229–263. https://doi.org/10.3322/caac.21834
[2] Houcek, A. J., Uzay, B., Ildefonso, G. V., Omokehinde, T., Monteggia, L. M., Quaranta, V., Linkous, A., & Kavalali, E. T. (2025). Small cell lung cancer induces synaptic scaling to alter neuronal excitability. Communications Biology, 8(1), 1559. https://doi.org/10.1038/s42003-025-09006-7
[3] Kim, S. Y., Park, H. S., & Chiang, A. C. (2025). Small Cell Lung Cancer: A Review. JAMA, 333(21), 1906–1917. https://doi.org/10.1001/jama.2025.0560
[4] Das, S., & Samaddar, S. (2025). Recent Advances in the Clinical Translation of Small-Cell Lung Cancer Therapeutics. Cancers, 17(2), 255. https://doi.org/10.3390/cancers17020255
[5] The U.S. Interagency Staff Group on Carcinogens. (1986). Chemical Carcinogens: A Review of the Science and Its Associated Principles. Environmental Health Perspectives, 67, 201–282. https://doi.org/10.2307/3430333
[6] Yuan, M., Zhao, Y., Arkenau, H.-T., Lao, T., Chu, L., & Xu, Q. (2022). Signal pathways and precision therapy of small-cell lung cancer. Sig Transduct Target Ther, 7(1), 187. https://doi.org/10.1038/s41392-022-01013-y
[7] Semenova, E. A., Nagel, R., & Berns, A. (2015). Origins, genetic landscape, and emerging therapies of small cell lung cancer. Genes & Development, 29(14), 1447–1462. https://doi.org/10.1101/gad.263145.115
[8] Quaranta, V., & Linkous, A. (2022). Organoids as a Systems Platform for SCLC Brain Metastasis. Frontiers in Oncology, 12. https://doi.org/10.3389/fonc.2022.881989
[9] Chiang, C.-L., Yang, H.-C., Liao, Y.-T., Luo, Y.-H., Wu, Y.-H., Wu, H.-M., & Chen, Y.-M. (2023). Treatment and survival of patients with small cell lung cancer and brain metastasis. Journal of Neuro-Oncology, 165(2), 343–351. https://doi.org/10.1007/s11060-023-04512-2
[10] Rittberg, R., Banerji, S., Kim, J. O., Rathod, S., & Dawe, D. E. (2021). Treatment and Prevention of Brain Metastases in Small Cell Lung Cancer. American Journal of Clinical Oncology, 44(12), 629. https://doi.org/10.1097/COC.0000000000000867
[11] Zhu, Y., Cui, Y., Zheng, X., Zhao, Y., & Sun, G. (2022). Small-cell lung cancer brain metastasis: From molecular mechanisms to diagnosis and treatment. Biochimica et Biophysica Acta: Molecular Basis of Disease, 1868(12), 166557–166557. https://doi.org/10.1016/j.bbadis.2022.166557
[12] Qu, F., Pasca, A., Kong, C., Winslow, M., & Sage, J. (2020). B23 Unraveling the Mechanisms of Small-Cell Lung Cancer Brain Metastasis. Journal of Thoracic Oncology, 15(2), S34. https://doi.org/10.1016/j.jtho.2019.12.089
[13] Steeg, P. S. (2021). The blood–tumour barrier in cancer biology and therapy. Nature Reviews Clinical Oncology, 18(11), 696–714. https://doi.org/10.1038/s41571-021-00529-6
[14] Onganer, P. U., Seckl, M. J., & Djamgoz, M. B. A. (2005). Neuronal characteristics of small-cell lung cancer. British Journal of Cancer, 93(11), 1197–1201. https://doi.org/10.1038/sj.bjc.6602857
[15] Simpson, K. L., Rothwell, D. G., Blackhall, F., & Dive, C. (2025). Challenges of small cell lung cancer heterogeneity and phenotypic plasticity. Nature Reviews Cancer, 25(6), 447–462. https://doi.org/10.1038/s41568-025-00803-0
[16] Park, S. J., Kim, Y. Y., Han, J. Y., Kim, S. W., Kim, H., & Ku, S.-Y. (2024). Advancements in Human Embryonic Stem Cell Research: Clinical Applications and Ethical Issues. Tissue Engineering and Regenerative Medicine, 21(3), 379–394. https://doi.org/10.1007/s13770-024-00627-3
[17] Peng, T., Ma, X., Hua, W., Wang, C., Chu, Y., Sun, M., Fermi, V., Hamelmann, S., Lindner, K., Shao, C., Zaman, J., Tian, W., Zhuo, Y., Harim, Y., Stöffler, N., Hammann, L., Xiao, Q., Jin, X., Warta, R., … Mao, Y. (2025). Individualized patient tumor organoids faithfully preserve human brain tumor ecosystems and predict patient response to therapy. Cell Stem Cell, 32(4), 652-669.e11. https://doi.org/10.1016/j.stem.2025.01.002
[18] Scattolin, D., Maso, A. D., Ferro, A., Frega, S., Bonanno, L., Guarneri, V., & Pasello, G. (2024). The emerging role of Schlafen-11 (SLFN11) in predicting response to anticancer treatments: Focus on small cell lung cancer. Cancer Treatment Reviews, 128, 102768. https://doi.org/10.1016/j.ctrv.2024.102768