Millions of people across the world are diagnosed with neurodegenerative diseases, where we see the large-scale destruction of brain cells called neurons (1). These diseases are also considered one of the leading causes of death in the United States and the world (2, 3). Among the hundreds of neurodegenerative diseases that have been characterized, Multiple Sclerosis (MS) is one of the most common – around 2.9 million people are currently diagnosed with MS (4), and there has been a significant increase in the last 30 years (5).
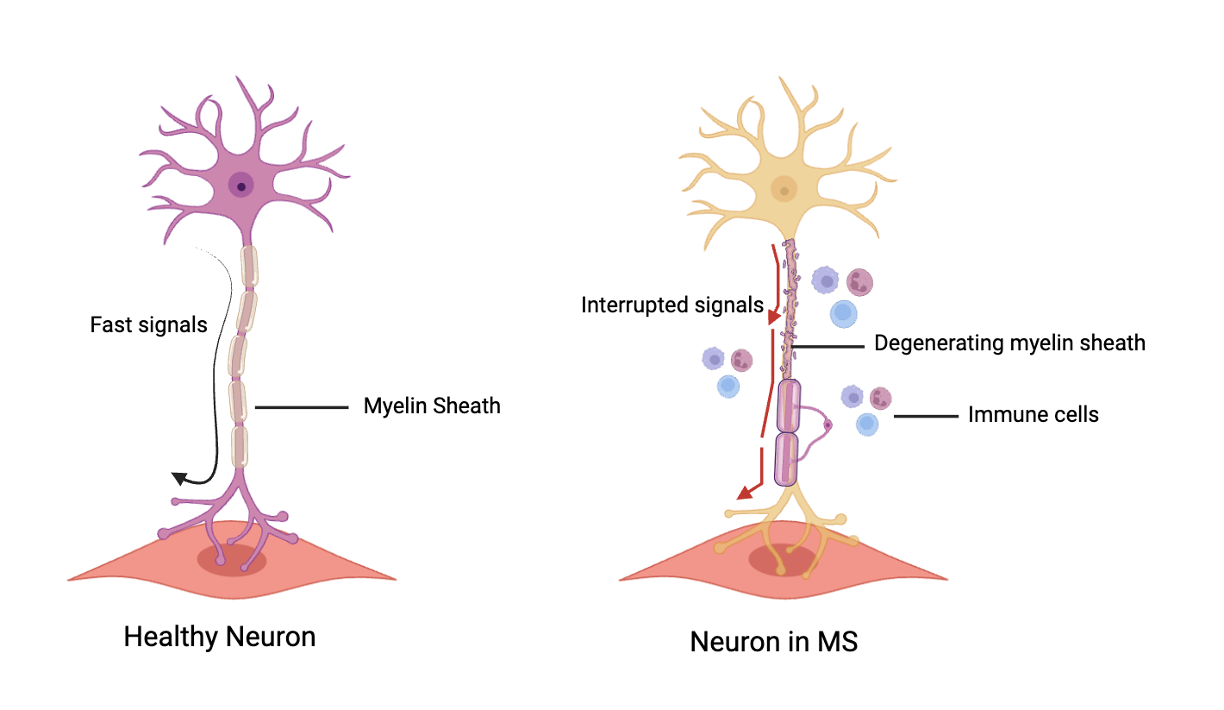
Our immune system, which is responsible for defending our body against infections, goes haywire in MS (6). Immune cells attack neurons in the brain and the spinal cord; more specifically, they attack the insulation that wraps around neurons (6). This insulation is important because it allows for electrical signals to flow quickly between neurons for communication, much like the insulation around electrical wires between the power grid and appliances at home. When this insulation 一 known as the myelin sheath 一 is significantly damaged, our neurons can no longer send reliable messages to each other (Figure 1) (7). Depending on where the immune system attacks neurons, we can see drastic symptoms arise, such as vision loss, loss of motor control, persistent nausea, impaired cognition, and widespread inflammation (6). Over time, these symptoms become worse, and while the time taken to progress to late-stage MS differs among patients (8), it is a fatal disease. All current drugs on the market focus on delaying the progression of MS (6). They do not stop or cure MS, but they can significantly improve the quality of life for the patient.
At first glance, to those slightly familiar with MS, the treatment seems straightforward: kill the rogue immune cells and prevent neuronal death! Many drugs do this by suppressing immune system activity by binding to overactive immune cells and preventing them from destroying the myelin sheath (6). However, these drugs are extremely expensive, and even if one could afford them, they are not enough to completely stop MS progression (6). This is because the damage the immune system causes is still largely irreversible: we do not yet know how to regenerate the myelin sheath around neurons. MS takes time to be diagnosed, which means that treatment starts midway into the immune system’s attack on neurons 一 we catch it too late and cannot prevent the early death of myelin. Suppressing the body’s immune system also leaves the patient susceptible to everyday diseases, such as the flu, pneumonia, or mild bacterial infections. Lastly, immune cells don’t just affect the myelin sheath on neurons; they also affect other cells that support the nervous system (9). While largely overlooked among neurons, these cells are crucial for maintaining a conducive and nourishing environment in the brain and spinal cord (10). Their loss and/or overactivity in MS causes further damage to the neurons by stressing them and eventually killing them (9). However, these cells may also hold the solution to MS treatment 一 instead of only focusing on treating our main culprit, the overactive immune system. Changing the properties of these supporting cells via drugs can help the neuron recover, and potentially, gain back the myelin sheath over time.
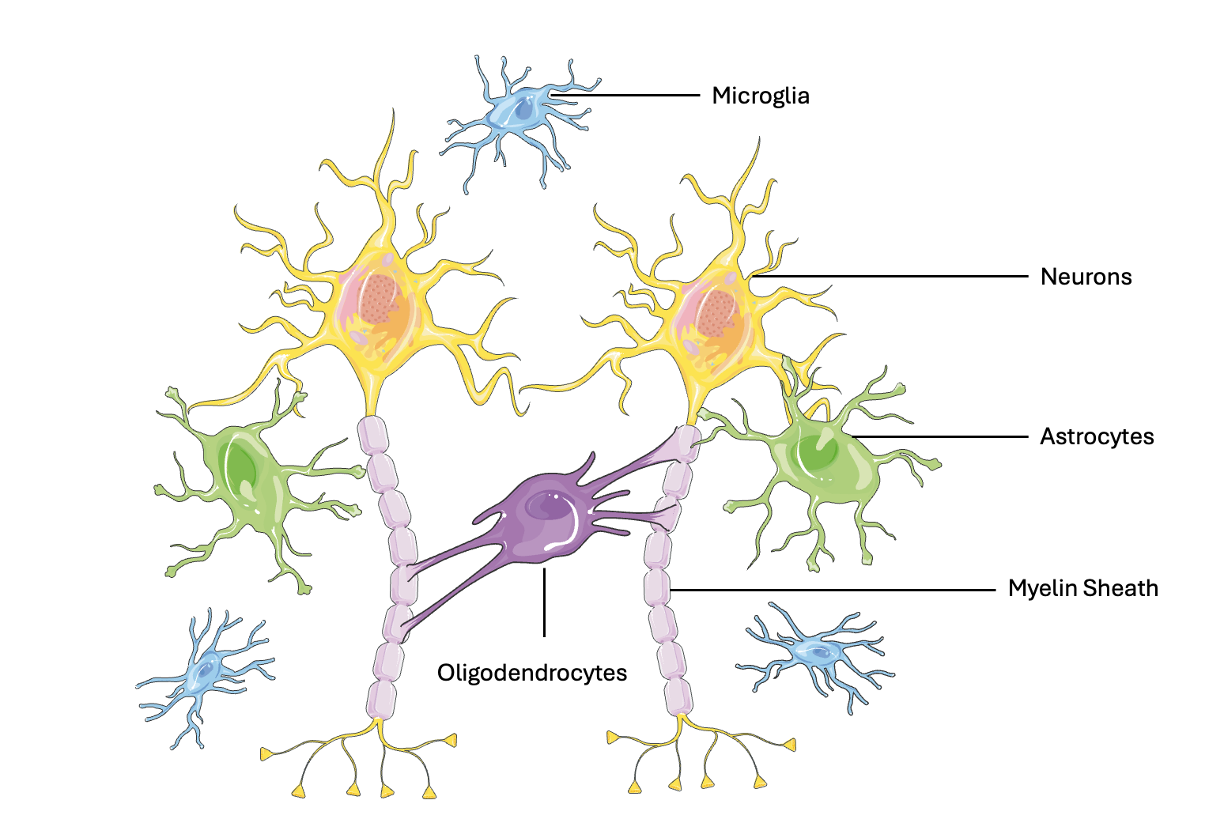
These supporting cells of the nervous system are called glia or glial cells (10), which make up half of all cells in the nervous system. Each glial cell type has its own specialized role in maintaining a healthy and functioning nervous system (Figure 2) (10). Three main glial cell types are important to understand MS progression and future treatment:
- Astrocytes: Like scaffolding for a building, astrocytes provide physical support for neurons and other glial cells (11). Neurons are extremely energy-hungry, and astrocytes ensure they get the essential nutrients they need. Astrocytes also clean up the remnants of messages sent between neurons, to ensure that the next message is clearly communicated (11).
- Oligodendrocytes: They are responsible for producing and maintaining the myelin sheath. They wrap around the neuron in sections, allowing the electrical signal to flow quickly (10).
- Microglia: They defend the nervous system. They patrol and assess neuronal functioning; if a neuron is unhealthy or damages other neurons, they attack it and prevent further harm (10).
To understand how we can get these glial cells to start protecting, nourishing, and supporting the nervous system in MS, we must first understand how they do so. The cells that make up nearly all life on Earth need energy to carry out important functions, like producing proteins, replicating to form more cells, and maintaining balance (12). Cells do this by utilizing sugar, or more specifically glucose, as an energy molecule by converting it to ATP (adenosine triphosphate) (12). ATP acts as the energy currency, which is used when cells perform functions to sustain the body (12).
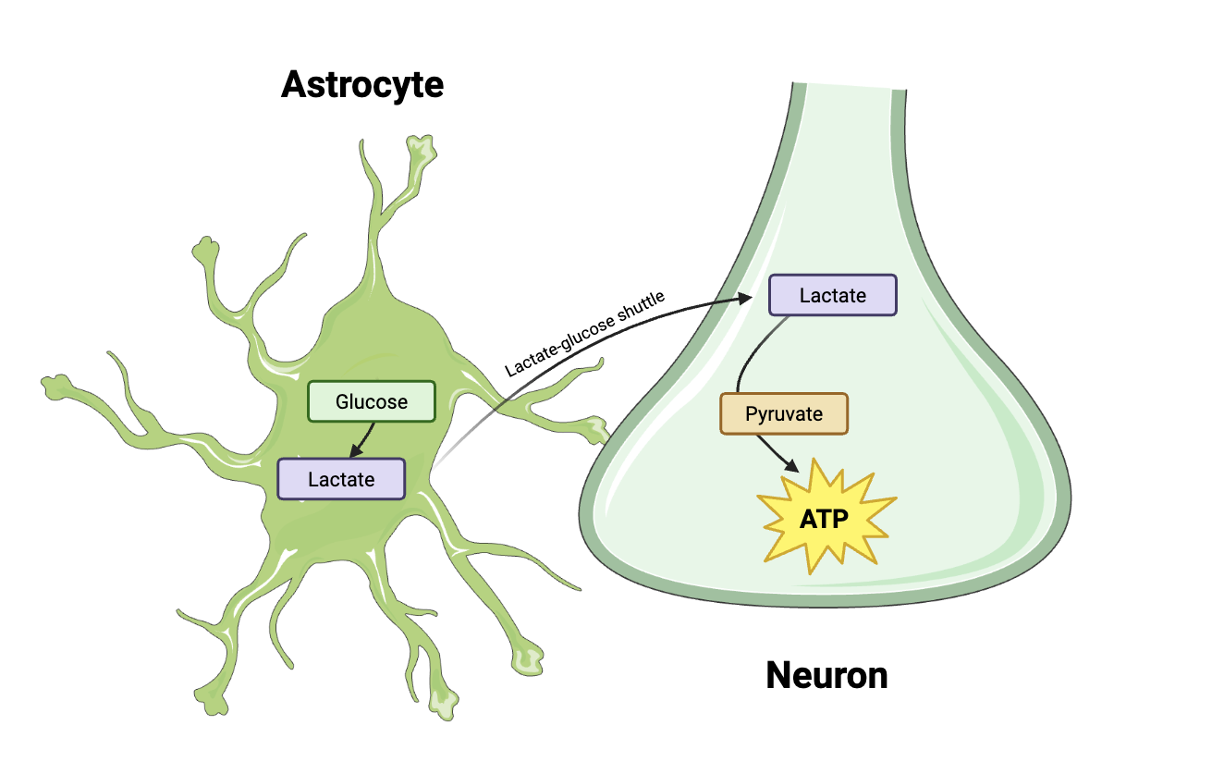
However, unlike other cells, neurons rely on different energy sources depending on what they do. During periods of high brain activity, they need lots of energy quickly, a demand that glucose cannot meet. Lactate, a molecule derived from glucose, is a more efficient fuel source in such scenarios. Neurons rapidly import lactate, quickly convert it to pyruvate, the precursor for ATP production (13, 14). The responsibility for supplying this critical lactate falls onto astrocytes. Astrocytes convert glucose into lactate and deliver it to neurons and other glial cells, thereby playing an essential role in sustaining the constant energy supply neurons demand during active signaling (13). How does lactate reach neurons and other glia? Astrocytes transport lactate via a shuttle (Figure 3) to neurons and glial cells, where the rapid uptake of lactate ensures a constant ATP supply (13).
The insights into these differences in energy production were the results of a technique known as NMR (Nuclear Magnetic Resonance Spectroscopy). In NMR, scientists specifically mark sugar molecules in glia and neurons by adding a tag, similar to adding collars on pets. They then place the brain cells in a very strong magnetic field. The end-products of the sugar molecules (here, lactate and glucose) are then measured via the level of tags in each cell. By analyzing the results, we can track the amount and type of sugars used to produce energy in astrocytes compared to neurons. Using this method solidified the important role that astrocytes take on to nourish neurons and other glia (14).
In MS, the immune cells that attack the myelin sheath also attack astrocytes, leading to an overactive state (11). This significantly disturbs the lactate-glucose shuttle system, preventing neurons and oligodendrocytes from meeting their energy requirements that could be used to help replenish the myelin sheath (15). Apart from disrupting energy flow, overactive astrocytes also release toxic molecules that directly scar and destroy the myelin sheath, oligodendrocytes, and the neurons themselves (11, 16). Astrocytes also lose their ability to maintain structural support, which is evident at the blood-brain barrier, a filter controlling entry into the brain. The loss of support leads to a breakdown of the cells that connect the filter, making the brain more susceptible to infections and other harmful foreign agents.
This trend of the immune system pushing helpful glia into overactive and harmful states in MS does not stop with astrocytes; it occurs with microglia as well. The main role of microglia lies in their ability to clear debris in the brain 一 from clumps of toxic buildup to dead and dying neurons and glia (10). One way microglia can clear this debris is by taking in these cells and converting their components into energy. When it comes to the myelin sheath on neurons, which is made up of fatty compounds called lipids, microglia do the same. They break the sheath into small fat droplets and have sensors that find these droplets and convert them into energy to sustain themselves (17).
Immune cells in MS cause dysfunctions in microglia by affecting the fat sensors that clear debris and remnants of the myelin sheath (18). This causes an accumulation of fat droplets around the neuron, which invites more immune cells to attack nearby neurons, as they cannot be cleared by microglia. This leads to a rapid cascade of even further degeneration and eventually the death of neurons. Alternatively, some immune cells can also overactivate microglia. Since they are capable of pruning the nervous system by removing dead and dying neurons and glia, overactive microglia go ‘rogue’ and start killing surrounding neurons, astrocytes, oligodendrocytes, and other glial cells, even when they are healthy (19), contributing to further neuronal loss in MS.
Current MS treatments largely focus on addressing immune cell reactivity, as immune cells are considered the main antagonists of the disease (6). However, focusing on the main ‘culprits’ may not be the best avenue to treat MS, as well as other neurodegenerative diseases. We must keep in mind that these diseases are rarely one-dimensional, and correcting only one aspect may not result in improvement. To treat multi-dimensional diseases like MS, doctors and scientists should consider focusing on other parts of the body that are going haywire. When we shift our therapeutic focus from ‘target the damaged component’ to also ‘target the supporting ecosystem,’ we open new doors to more effective treatments. Rather than asking ‘How do we kill the harmful cells (here, immune cells)?’, we can instead focus on a perspective that is largely underrepresented in medical research 一 How do we help the supportive cells help the damaged cells? For MS, we must consider glial therapy alongside immunotherapy as a potential avenue to give the myelin sheath a chance to regenerate.
Indeed, current research has shown that changing glial function in MS can help myelin regeneration. Studies have shown that preventing the overactivation of astrocytes and microglia by blocking their interactions with immune cells lowers inflammation (16), and clinical trials with this ‘glial-cell blocker’ have shown that 80% of patients with MS do not relapse in 2 years (20). Currently, clinical trials to prevent astrocytes from releasing toxic compounds are underway (21). While these strategies base their effectiveness on targeting the detrimental effects of glia, those that enhance their nourishing and protective functions could be a better alternative or more effective if combined with glial-cell blockers and immunotherapy.
Researchers are presently investigating how changing astrocytic and microglial support when it comes to metabolism of glucose and lipids, respectively, can address remyelination in MS. Helping astrocytes shuttle energy to oligodendrocytes via glucose and lipids has been shown to regulate remyelination (22). Changing the levels of fat sensors in overactive and under-reactive microglia to bring them to a more balanced state has improved the repair of the myelin sheath in cells cultured in the lab (18). Enhancing the transport of lactate from astrocytes to neurons has been shown to meet increased energy demands that are typically seen in MS (23). Considering that lactate is a neuroprotective molecule as well (24), it is then easy to see why changing the energy pathways of astrocytes in neurons is beneficial for those with MS. Lactate is currently being used as a supplementary treatment for those with traumatic brain injury (25), but primary research is still underway to investigate how the glucose-lactate shuttle can be targeted in MS (23).
Glia have been overlooked for many years in biomedical research, but as we have seen with MS, focusing on these cells will allow scientists and doctors to create effective treatments for a disease that is largely fatal. By shifting our perspective, new treatment avenues can be created not only for MS, but for other neurodegenerative diseases like Alzheimer’s Disease, Parkinson’s Disease, and Frontotemporal Dementia, among many others. This can extend to other organs and cell types that have been overlooked, like glia, when it comes to diseases that are inherently hard to completely cure, like diabetes, heart disease, kidney disease, and so on. Glia and other supporting cells in our body are not background players—they are essential and could be considered as ‘underdogs’. Recognizing their importance and developing therapies that support them may be the key to finally treating the diseases that have resisted effective treatment for decades. The future of treatment lies not in ignoring these cells, but in using their functions to give them the capacity to heal.
References:
- Przedborski, S., Vila, M., & Jackson-Lewis, V. (2003). Series Introduction: Neurodegeneration: What is it and where are we? Journal of Clinical Investigation, 111(1), 3–10. https://doi.org/10.1172/jci17522
- Ahmad, F. B., & Anderson, R. N. (2021). The leading causes of death in the US for 2020. JAMA, 325(18), 1829. https://doi.org/10.1001/jama.2021.5469
- Naghavi, M., Ong, K. L., Aali, A., Ababneh, H. S., Abate, Y. H., Abbafati, C., Abbasgholizadeh, R., Abbasian, M., Abbasi-Kangevari, M., Abbastabar, H., ElHafeez, S. A., Abdelmasseh, M., Abd-Elsalam, S., Abdelwahab, A., Abdollahi, M., Abdollahifar, M., Abdoun, M., Abdulah, D. M., Abdullahi, A., . . . Al-Aly, Z. (2024). Global burden of 288 causes of death and life expectancy decomposition in 204 countries and territories and 811 subnational locations, 1990–2021: a systematic analysis for the Global Burden of Disease Study (2021). The Lancet, 403(10440), 2100–2132. https://doi.org/10.1016/s0140-6736(24)00367-2
- Number of people with MS | Atlas of MS. (2026). https://atlasofms.org/map/global/epidemiology/number-of-people-with-ms
- Koch-Henriksen, N., & Magyari, M. (2021). Apparent changes in the epidemiology and severity of multiple sclerosis. Nature Reviews Neurology, 17(11), 676–688. https://doi.org/10.1038/s41582-021-00556-y
- Boutitah-Benyaich, I., Eixarch, H., Villacieros-Álvarez, J., Hervera, A., Cobo-Calvo, Á., Montalban, X., & Espejo, C. (2025). Multiple sclerosis: molecular pathogenesis and therapeutic intervention. Signal Transduction and Targeted Therapy, 10(1), 324. https://doi.org/10.1038/s41392-025-02415-4
- Popescu, B. F., & Lucchinetti, C. F. (2011). Pathology of demyelinating diseases. Annual Review of Pathology Mechanisms of Disease, 7(1), 185–217. https://doi.org/10.1146/annurev-pathol-011811-132443
- Lublin, F. D., Reingold, S. C., Cohen, J. A., Cutter, G. R., Sørensen, P. S., Thompson, A. J., Wolinsky, J. S., Balcer, L. J., Banwell, B., Barkhof, F., Bebo, B., Calabresi, P. A., Clanet, M., Comi, G., Fox, R. J., Freedman, M. S., Goodman, A. D., Inglese, M., Kappos, L., . . . Polman, C. H. (2014). Defining the clinical course of multiple sclerosis. Neurology, 83(3), 278–286. https://doi.org/10.1212/wnl.0000000000000560
- Barnett, M. H., & Prineas, J. W. (2004). Relapsing and remitting multiple sclerosis: Pathology of the newly forming lesion. Annals of Neurology, 55(4), 458–468. https://doi.org/10.1002/ana.20016
- Allen, N. J., & Lyons, D. A. (2018). Glia as architects of central nervous system formation and function. Science, 362(6411), 181–185. https://doi.org/10.1126/science.aat0473
- Correale, J., & Farez, M. F. (2015). The role of astrocytes in multiple sclerosis progression. Frontiers in Neurology, 6, 180. https://doi.org/10.3389/fneur.2015.00180
- Bonora, M., Patergnani, S., Rimessi, A., De Marchi, E., Suski, J. M., Bononi, A., Giorgi, C., Marchi, S., Missiroli, S., Poletti, F., Wieckowski, M. R., & Pinton, P. (2012). ATP synthesis and storage. Purinergic Signalling, 8(3), 343–357. https://doi.org/10.1007/s11302-012-9305-8
- Brown, A. M., Tekkök, S. B., & Ransom, B. R. (2004). Energy transfer from astrocytes to axons: the role of CNS glycogen. Neurochemistry International, 45(4), 529–536. https://doi.org/10.1016/j.neuint.2003.11.005
- Pellerin, L., Pellegri, G., Bittar, P. G., Charnay, Y., Bouras, C., Martin, J., Stella, N., & Magistretti, P. J. (1998). Evidence supporting the existence of an Activity-Dependent Astrocyte-Neuron lactate shuttle. Developmental Neuroscience, 20(4–5), 291–299. https://doi.org/10.1159/000017324
- Korn, T., Magnus, T., & Jung, S. (2005). Autoantigen specific T cells inhibit glutamate uptake in astrocytes by decreasing expression of astrocytic glutamate transporter GLAST: a mechanism mediated by tumor necrosis factor‐α. The FASEB Journal, 19(13), 1878–1880. https://doi.org/10.1096/fj.05-3748fje
- Ortiz, C. C., & Eroglu, C. (2023). Astrocyte signaling and interactions in Multiple Sclerosis. Current Opinion in Cell Biology, 86, 102307. https://doi.org/10.1016/j.ceb.2023.102307
- Chausse, B., Kakimoto, P. A., & Kann, O. (2020). Microglia and lipids: how metabolism controls brain innate immunity. Seminars in Cell and Developmental Biology, 112, 137–144. https://doi.org/10.1016/j.semcdb.2020.08.001
- Dong, Y., D’Mello, C., Pinsky, W., Lozinski, B. M., Kaushik, D. K., Ghorbani, S., Moezzi, D., Brown, D., Melo, F. C., Zandee, S., Vo, T., Prat, A., Whitehead, S. N., & Yong, V. W. (2021). Oxidized phosphatidylcholines found in multiple sclerosis lesions mediate neurodegeneration and are neutralized by microglia. Nature Neuroscience, 24(4), 489–503. https://doi.org/10.1038/s41593-021-00801-z
- Yong, V. W. (2022). Microglia in multiple sclerosis: Protectors turn destroyers. Neuron, 110(21), 3534–3548. https://doi.org/10.1016/j.neuron.2022.06.023
- Vermersch, P., Airas, L., Berger, T., Deisenhammer, F., Grigoriadis, N., Hartung, H., Magyari, M., Popescu, V., Pozzilli, C., Pugliatti, M., Van Wijmeersch, B., Zakaria, M., & Oreja-Guevara, C. (2025). The role of microglia in multiple sclerosis: implications for treatment with Bruton’s tyrosine kinase inhibitors. Frontiers in Immunology, 16, 1495529. https://doi.org/10.3389/fimmu.2025.1495529
- Colombo, E., Bassani, C., De Angelis, A., Ruffini, F., Ottoboni, L., Comi, G., Martino, G., & Farina, C. (2020). Siponimod (BAF312) activates NRF2 while hampering NFΚB in human astrocytes, and protects from Astrocyte-Induced neurodegeneration. Frontiers in Immunology, 11, 635. https://doi.org/10.3389/fimmu.2020.00635
- Molina-Gonzalez, I., Holloway, R. K., Jiwaji, Z., Dando, O., Kent, S. A., Emelianova, K., Lloyd, A. F., Forbes, L. H., Mahmood, A., Skripuletz, T., Gudi, V., Febery, J. A., Johnson, J. A., Fowler, J. H., Kuhlmann, T., Williams, A., Chandran, S., Stangel, M., Howden, A. J. M., . . . Miron, V. E. (2023). Astrocyte-oligodendrocyte interaction regulates central nervous system regeneration. Nature Communications, 14(1), 3372. https://doi.org/10.1038/s41467-023-39046-8
- Liu, Z., Guo, Y., Zhang, Y., Gao, Y., & Ning, B. (2024). Metabolic reprogramming of astrocytes: Emerging roles of lactate. Neural Regeneration Research, 21(2), 421–432. https://doi.org/10.4103/nrr.nrr-d-24-00776
- Newington, J. T., Harris, R. A., & Cumming, R. C. (2013). Reevaluating Metabolism in Alzheimer’s Disease from the Perspective of the Astrocyte-Neuron Lactate Shuttle Model. Journal of Neurodegenerative Diseases, 2013, 1–13. https://doi.org/10.1155/2013/234572
- Bhatti, M. S., & Frostig, R. D. (2023). Astrocyte-neuron lactate shuttle plays a pivotal role in sensory-based neuroprotection in a rat model of permanent middle cerebral artery occlusion. Scientific Reports, 13(1), 12799. https://doi.org/10.1038/s41598-023-39574-9