Introduction: Context and Tension
Imagine you are at a summer music festival, shoulder-to-shoulder with thousands of people, when someone nearby leans over and offers you “magic mushrooms”. In that setting, the societal perception and cultural story you know is clear: psychedelic mushrooms are dangerous, illegal, and medically useless.
However, if that same person handed you a prescription bottle with a synthesized drug rather than a plastic baggie filled with dry mushrooms, and you were not at a concert venue but in a clinic with trained medical professionals, you could be looking at an FDA-designated breakthrough therapy for treatment-resistant depression and major depressive disorder (MDD).
This contrast captures the central tension surrounding psychedelic mushrooms’ place in medicine. Psilocybin, the main psychoactive compound in what is commonly known as magic mushrooms, is federally classified in the United States as a Schedule 1 drug by the U.S. Drug Enforcement Administration. This category is reserved for substances that the Drug Enforcement Administration has labeled as having “no currently accepted medical use” and a “high potential for abuse” [1]. By this definition, psilocybin sits alongside drugs like heroin, treated as maximally dangerous and minimally useful.
Yet, at the same time, the U.S. Food and Drug Administration has designated psilocybin-assisted therapy as a breakthrough therapy for treatment-resistant depression and MDD [2, 3]. This status reflects findings from multiple clinical trials showing large, rapid, and often persisting reductions in depressive symptoms in tandem with psilocybin-assisted therapy [4, 5].
This conflict between different official governmental institutions and emergent data is the central issue addressed in this article, as modern neuroscience is beginning to show that psilocybin may have therapeutic value despite its Schedule 1 classification.
Through carefully guided, monitored sessions, psilocybin may temporarily shake up our brains like a snow globe, disrupting rigid brain patterns of thinking, behavior, and emotion, helping individuals escape habitually negative thought patterns so new, healthier patterns and habits of thought and behavior can form [6, 7, 8]. Imaging technology, cellular studies, and longitudinal clinical trials are converging on the idea that psilocybin induces a brief period of increased brain reorganization, particularly in systems related to self-referential thinking, emotion, and stress, which may contribute to reduced depressive symptoms [4, 5, 6, 7, 8].
Despite growing evidence, currently, societal perception of the context in which psychedelic mushrooms are used is recreational, non-clinical, and degenerate. U.S. drug scheduling decisions made in the early 1970s froze psilocybin in place legally, socially, and medically. This classification has made it extremely difficult to study psilocybin’s potential therapeutic effects, leaving many important gaps in our understanding of this substance and creating stigma.
To further exacerbate this issue, conventional therapies for MDD, such as selective serotonin reuptake inhibitors (SSRIs) and weekly talk therapy, can be life-changing for some people, but leave many others only partially relieved, facing side effects, cycling through multiple different therapists, or requiring ongoing daily doses of drugs over many years to maintain relief [9, 10].
Psilocybin-assisted therapy offers a radically different model for depression therapy: a few carefully prepared, monitored, and guided sessions that can produce sustained improvements in mood and quality of life months after the drug has been cycled from the body [4, 5, 11].
Psilocybin and Its Mechanism of Action
Psilocybin is a psychoactive, hallucinogenic compound found in more than 100 species of mushrooms that humans have ingested and used in spiritual and healing contexts across centuries [4]. After ingestion, psilocybin is converted into a different molecule called psilocin [6]. Psilocin can cross a highly selective “security system” that carefully regulates which substances are allowed into the brain, called the blood-brain barrier [12].
Psilocin itself acts like a key that fits a particular lock called a receptor—specifically, the serotonin 5-HT2A receptor, which can be found on the surface of neurons [13]. Usually, this type of lock is activated by serotonin, a natively produced signaling chemical involved in mood, sleep, appetite, and social behavior [13]. When serotonin binds to a 5-HT2A receptor, it triggers a chain of internal signals inside the neuron that change how strongly that neuron signals to other neurons [13]. Sometimes the signal strengthens connections between neurons; other times it weakens them [13, 14]. This flexible rewiring of connections is one way the brain demonstrates neuroplasticity—its capacity to reorganize its structure and function over time [6, 7].
Psilocin can bind to the same lock as serotonin—5-HT2A receptors—but produces a slightly different cascade of signaling effects than natural serotonin [13]. In animal studies and more cellular studies, when psilocin “unlocks” 5-HT2A receptors, signaling pathways that support the growth of new neuronal connections, and the health of existing ones, are activated—a process known as synaptogenesis, or the formation of connections between neurons, which is a marker of increased neuroplasticity and improved brain health [6, 7, 14].
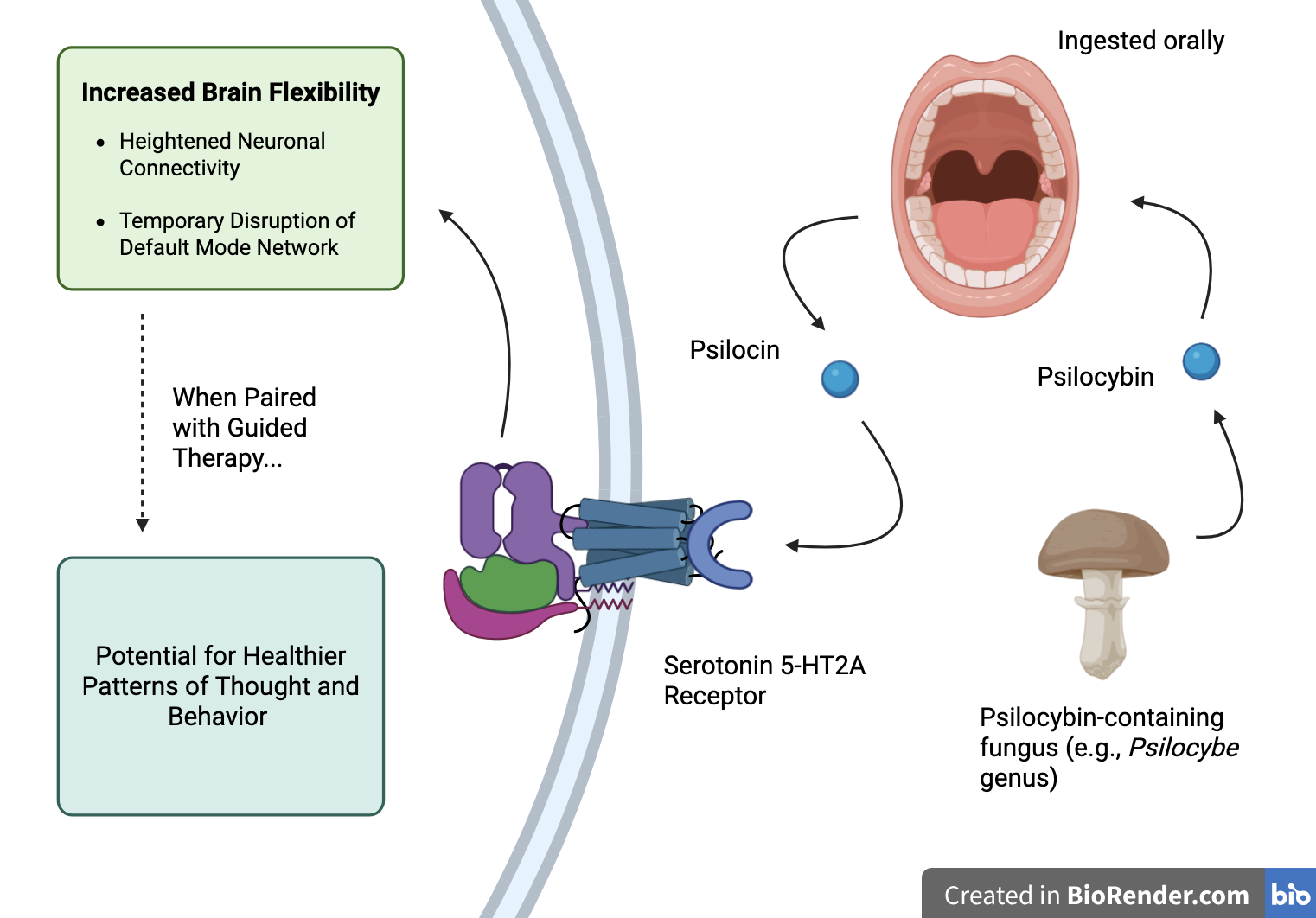
Figure 1: Proposed pathway of psilocybin’s effects: psilocybin-containing fungi are ingested orally, psilocybin is converted to psilocin, psilocin unlocks serotonin 5-HT2A receptors which increases neural flexibility that may support healthier thought patterns and behavior when paired with guided therapy.
5-HT2A receptors are most dense in the gut and in specific areas of the brain that are all part of a large-scale network called the default mode network (DMN), which becomes active when your mind is not focused on the outside world; for example, when you are daydreaming, thinking about yourself, or replaying memories of past events [15, 16, 17]. The DMN is made up of the prefrontal cortex (Figure 2a), which is a brain region generally involved in planning, decision making, and self-reflection, the posterior cingulate cortex (Figure 2b), which is generally involved in internally directed thought, self-reflection, and memory-related awareness, and other related regions [17].
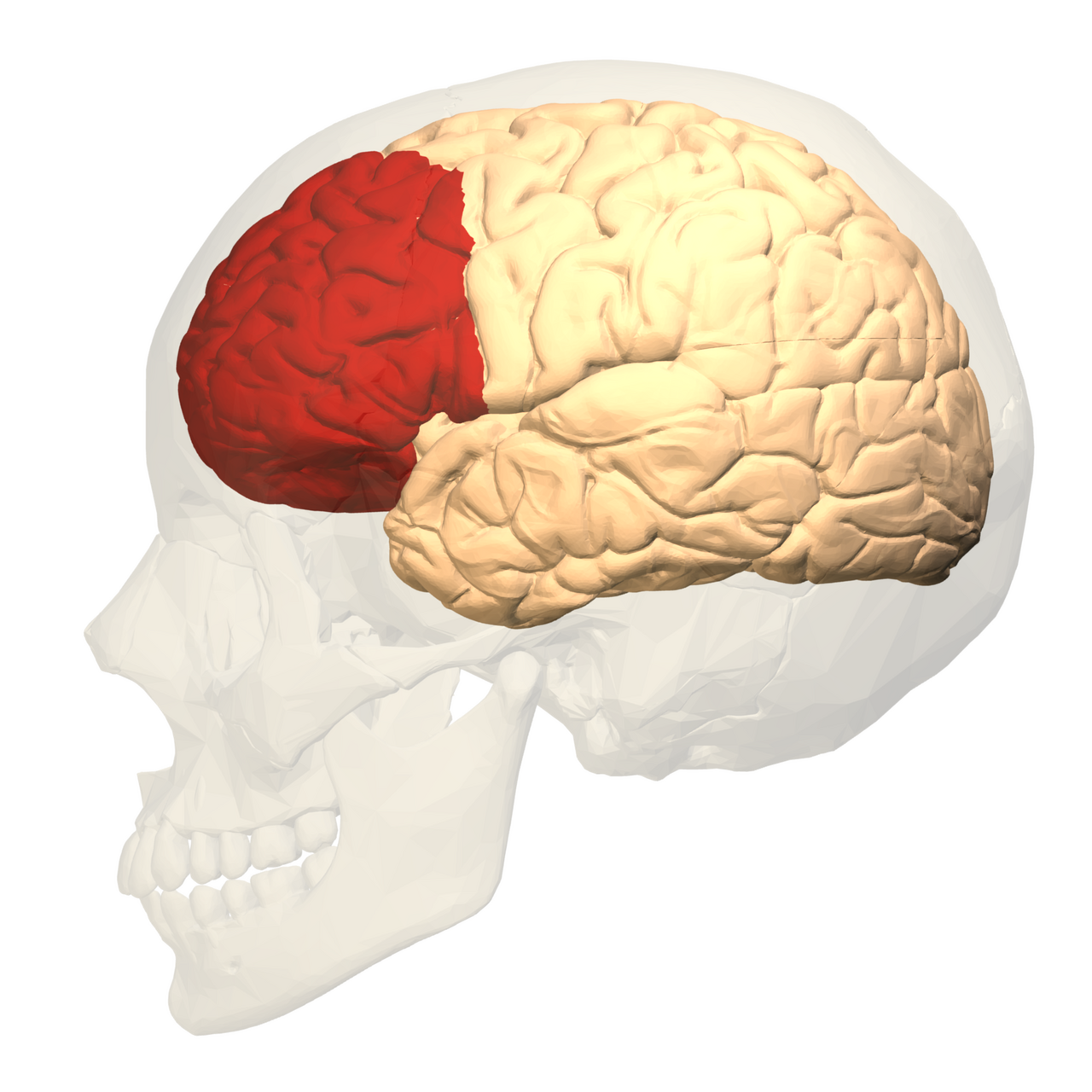
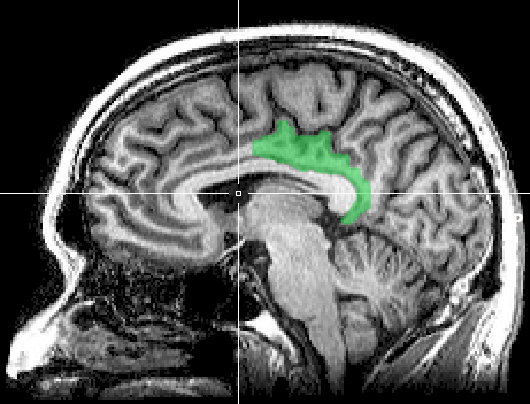
Figure 2: Depictions of two brain regions involved in the DMN: (a, on the left) lateral view of the prefrontal cortex, and (b, on the right) medial view of the posterior cingulate cortex.
In depressed individuals, the DMN typically becomes over-connected and rigid, predisposing people to repetitive loops of self-critical, hopeless thoughts often known as rumination [17]. By binding to 5-HT2A receptors in the DMN, psilocin appears to temporarily disrupt these rigid patterns and activate a window of increased neuroplasticity [8, 17].
One way to visualize the DMN is to picture the brain as a snow globe. Over years of experiences, habits, and stress, the snow settles into stable grooves—repetitive patterns of thought, emotion, and behavior. In depression, addiction, and chronic stress, many of these patterns are detrimental, pulling the mind back into the same loops of hopelessness, craving, or fear. Psilocybin acts like a strong shake of the snow globe: for a brief period, the habitual patterns are disrupted, and the snow swirls freely before settling again.
At the cellular level, this shake is a burst of reorganization—psilocin “unlocks” 5-HT2A receptors, triggering a domino effect of signals that support the growth of new neuronal connections, which promotes a potential escape from habitual lines of thought and rumination. In other words, under a psilocybin trip, the brain temporarily becomes more plastic, malleable, and open, increasing its capacity to generate new, healthier patterns of connectivity that can persist long after the snow has settled.
Research Techniques: Wait, How Do We Know This?
To understand the effects and mechanisms of psilocybin in the brain, neuroscientists have combined various techniques, including brain imaging and clinical trials [4, 17].
In human participants, imaging techniques, such as functional magnetic resonance imaging (fMRI), are used to track how brain regions activate before, after, and sometimes during a psilocybin trip [12, 17]. fMRI specifically measures changes in blood flow and oxygen use across different brain regions, which are markers of neural activity—this is how we know that the prefrontal cortex and posterior cingulate cortex are part of the DMN [15,17]. Imaging studies consistently demonstrate that psilocybin desynchronizes rigid patterns of connectivity within the DMN and related networks, supporting the running snow globe analogy [8, 17].
Clinical trials help connect these changes in brain connectivity to symptoms. In some studies, participants receive either psilocybin or a placebo, alongside guided therapy [5, 11]. Researchers use standardized scales like depression inventories measuring symptom severity, self-report alcohol usage, and structured interviews for trauma-related symptoms to measure participants’ levels of depression, substance use, and post-traumatic stress disorder (PTSD) over time [5, 11]. By comparing symptoms before and after treatment, we can begin to understand how the cellular changes under a psilocybin trip contribute to behavior and mental health.
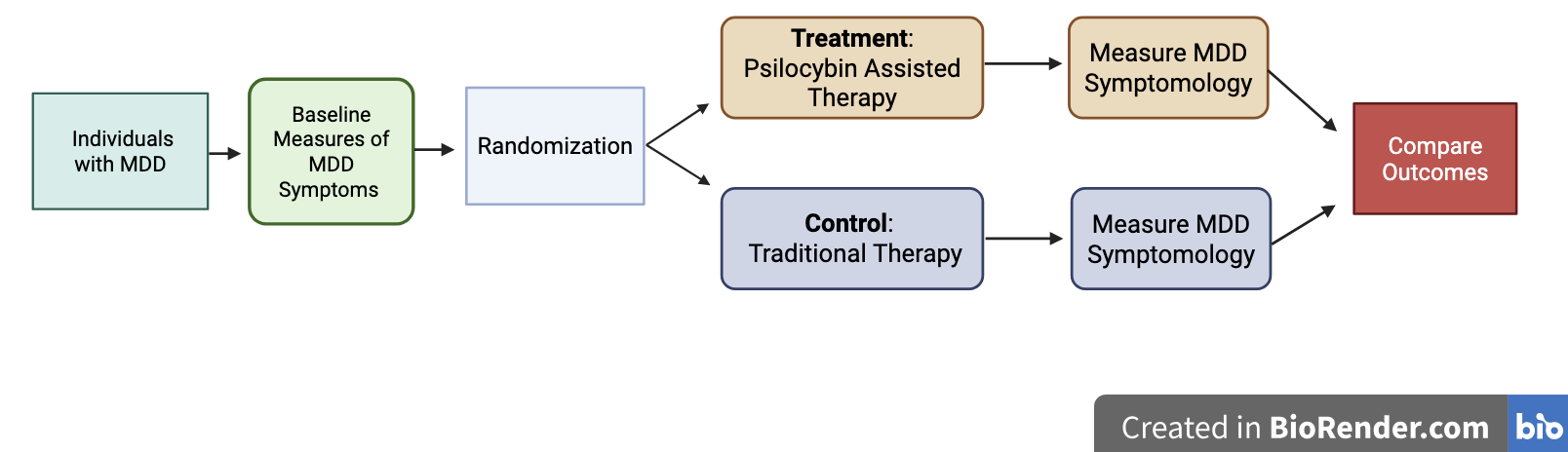
Figure 3: Proposed clinical trial design used to evaluate psilocybin-assisted therapy for major depressive disorder, showing baseline symptom assessment, randomization, treatment or control conditions, follow-up symptom measurement, and comparison of outcomes.
Psilocybin-Assisted Therapy: Depression, Substance Abuse, and Stress
A growing body of clinical work has focused on psilocybin-assisted therapy for MDD, including in individuals whose symptoms have not responded to standard treatments [4, 11]. In one study, adults with MDD received two psilocybin sessions combined with guided therapy [5]. Researchers found that depression scores dropped significantly after the first session and remained significantly improved four weeks after the second session [5]. Many participants’ depressive symptom scores moved from the severe or moderate range to the mild or minimal range after psilocybin-assisted therapy [5]. Similar conclusions have been reached throughout the literature: across multiple trials, psilocybin-assisted therapy produces large and often sustained reductions in depressive symptoms [4, 11].
These clinical findings correspond with the mechanistic explanation discussed earlier: by temporarily disrupting rigid default mode network patterns and increasing neuroplasticity across specific pathways, psilocybin appears to generate a brief window in which integrated negative self-beliefs and habits can be re-assessed, overcome, and updated in the context of supportive therapy [6, 8, 17]. Remember our snow globe analogy: depression is like snow that has settled, representing a rigid, self-critical pattern, while psilocybin shakes that snow loose and, with careful therapeutic guidance, can help it settle into healthier behaviors and patterns.
Psilocybin-assisted therapy has also been suggested as potentially promising in the treatment of substance use disorders, where habits of craving are supported by networks that include the DMN [12, 17]. Psilocybin, when strategically incorporated with structured therapy, can help reduce and hopefully even stop problematic use of substances like alcohol and nicotine [17, 18].
A similar logic surrounding the potential therapeutic effect of psilocybin is now being applied to stress-related and trauma-related conditions. Psilocybin and other psychedelics disrupt DMN activity, potentially making it easier for people to revisit and overcome painful memories with less fear and more openness [17].
While the effect of psilocybin-assisted therapy on depression is well supported, research on the effectiveness of psilocybin-assisted therapy on substance abuse and PTSD remains preliminary. Even so, the emergent pattern suggests that psilocybin-assisted therapy may offer a meaningful new option for people whose symptoms, across various conditions, have not fully responded to conventional treatments [4, 11, 18].
Is Psilocybin Really “Highly Addictive”?
The Schedule 1 label does not only claim that the substance has no accepted medical use—which has thus far been argued against—it also claims that the substance has a high potential for abuse [1, 19]. Classic psychedelics are often not associated with compulsive daily use, and people do not usually develop tolerance and withdrawal symptoms in the way that has been well documented with alcohol, opioids, and nicotine [20]. The literature generally emphasizes that psilocybin has a low potential for physiological dependence, no toxicity at doses used in clinical research, and limited evidence of problematic use patterns [4, 20].
It is essential to note that adverse psychological reactions such as anxiety, confusion, or psychotic-like symptoms can be associated with psychedelic use; however, this is most common in unsupervised settings and with individuals with personal or family histories of psychosis [13]. Considering these adverse psychological consequences, modern clinical studies use careful screening, guided therapy, and close monitoring [5, 11].
Importantly, the classic addiction criteria, including tolerance, withdrawal, and compulsive drug use, are not associated with psilocybin use [20]. The tension between the legal label of psilocybin as having a “high potential for abuse” and the emerging clinical evidence makes one question: Does psilocybin truly belong in the same category as heroin?
Schedule 1 Doesn’t Fit: Moving Forward
The U.S. Drug Enforcement Administration’s Schedule 1 classification is reserved for drugs that have no current accepted medical use and a high potential for abuse—the research reviewed suggests that psilocybin, when used in carefully controlled clinical settings, does not meet either of those criteria [1].
Reconsidering psilocybin’s current Schedule 1 designation would not mean a world where magic mushrooms are an unregulated lifestyle or wellness product; rather, it would allow for increased, more rigorous research on the topic, make it easier for clinicians and researchers to build safe treatment protocols for psilocybin-assisted therapy, and give people who have not found relief with conventional treatments access to emergent, potentially therapeutic interventions.
Returning to the crowded music festival: the key question is not whether it is wise to accept mushrooms from a stranger—that is highly dangerous and illegal—rather, the question is whether our laws should continue to treat the drug as if it had no legitimate medical value. The true challenge is to bring policy and public perception into alignment with evidence that psilocybin-assisted therapy may significantly improve the quality of life of many individuals with depression, substance abuse disorders, and trauma- or stress-related disorders.
Literature Cited:
- U.S. Drug Enforcement Administration. (2025, January 25). Psilocybin. https://www.dea.gov/factsheets/psilocybin
- U.S. Drug Enforcement Administration. (n.d.). Drug scheduling. https://www.dea.gov/drug-information/drug-scheduling.
- COMPASS Pathways. (2018). COMPASS Pathways receives FDA breakthrough therapy designation for psilocybin therapy for treatment‑resistant depression [Press release]. PR Newswire. https://www.prnewswire.com/news-releases/compass-pathways-receives-fda-breakthrough-therapy-designation-for-psilocybin-therapy-for-treatment-resistant-depression-834088100.html
- Agin-Liebes, G., & Davis, A. K. (2022). Psilocybin for the Treatment of Depression: A Promising New Pharmacotherapy Approach. Current Topics in Behavioral Neurosciences, 56, 125–140. https://doi.org/10.1007/7854_2021_282
- Davis, A. K., Barrett, F. S., May, D. G., Cosimano, M. P., Sepeda, N. D., Johnson, M. W., Finan, P. H., & Griffiths, R. R. (2021). Effects of Psilocybin-Assisted Therapy on Major Depressive Disorder: A Randomized Clinical Trial. JAMA Psychiatry, 78(5), 481–489. https://doi.org/10.1001/jamapsychiatry.2020.3285
- Fereydouni, S. (2025). Psilocybin and Neuroplasticity: A Review of Preclinical and Clinical Studies. OPEN Foundation. https://open-foundation.org/psilocybin-and-neuroplasticity/
- Grieco, S. F., Castrén, E., Knudsen, G. M., Kwan, A. C., Olson, D. E., Zuo, Y., Holmes, T. C., & Xu, X. (2022). Psychedelics and Neural Plasticity: Therapeutic Implications. The Journal of Neuroscience, 42(45), 8439–8449. https://doi.org/10.1523/JNEUROSCI.1121-22.2022
- Siegel, J. S., Subramanian, S., Perry, D., Kay, B. P., Gordon, E. M., Laumann, T. O., Reneau, T. R., Metcalf, N. V., Chacko, R. V., Gratton, C., Horan, C., Krimmel, S. R., Shimony, J. S., Schweiger, J. A., Wong, D. F., Bender, D. A., Scheidter, K. M., Whiting, F. I., Padawer-Curry, J. A., Dosenbach, N. U. F. (2024). Psilocybin desynchronizes the human brain. Nature, 632(8023), 131–138. https://doi.org/10.1038/s41586-024-07624-5
- Al-Harbi, K. S. (2012). Treatment-resistant depression: Therapeutic trends, challenges, and future directions. Patient Preference and Adherence, 6, 369–388. https://doi.org/10.2147/PPA.S29716
- Ruelaz, A. R. (2006). Treatment-Resistant Depression: Strategies for Management. Psychiatric Times, 23(11).
- Haikazian, S., Chen-Li, D. C. J., Johnson, D. E., Fancy, F., Levinta, A., Husain, M. I., Mansur, R. B., McIntyre, R. S., & Rosenblat, J. D. (2023). Psilocybin-assisted therapy for depression: A systematic review and meta-analysis. Psychiatry Research, 329, 115531. https://doi.org/10.1016/j.psychres.2023.115531
- Kwan, A. C., Olson, D. E., Preller, K. H., & Roth, B. L. (2022). The neural basis of psychedelic action. Nature Neuroscience, 25(11), 1407–1419. https://doi.org/10.1038/s41593-022-01177-4
- López-Giménez, J. F., & González-Maeso, J. (2018). Hallucinogens and Serotonin 5-HT2A Receptor-Mediated Signaling Pathways. Current Topics in Behavioral Neurosciences, 36, 45–73. https://doi.org/10.1007/7854_2017_478
- Raval, N. R., Johansen, A., Donovan, L. L., Ros, N. F., Ozenne, B., Hansen, H. D., & Knudsen, G. M. (2021). A Single Dose of Psilocybin Increases Synaptic Density and Decreases 5-HT2A Receptor Density in the Pig Brain. International Journal of Molecular Sciences, 22(2), 835. https://doi.org/10.3390/ijms22020835
- Andrews-Hanna, J. R., Smallwood, J., & Spreng, R. N. (2014). The default network and self-generated thought: Component processes, dynamic control, and clinical relevance. Annals of the New York Academy of Sciences, 1316(1), 29–52. https://doi.org/10.1111/nyas.12360
- Jakab, R. L., & Goldman-Rakic, P. S. (1998). 5-Hydroxytryptamine2A serotonin receptors in the primate cerebral cortex: Possible site of action of hallucinogenic and antipsychotic drugs in pyramidal cell apical dendrites. Proceedings of the National Academy of Sciences, 95(2), 735–740. https://doi.org/10.1073/pnas.95.2.735
- Gattuso, J. J., Perkins, D., Ruffell, S., Lawrence, A. J., Hoyer, D., Jacobson, L. H., Timmermann, C., Castle, D., Rossell, S. L., Downey, L. A., Pagni, B. A., Galvão-Coelho, N. L., Nutt, D., & Sarris, J. (2022). Default Mode Network Modulation by Psychedelics: A Systematic Review. International Journal of Neuropsychopharmacology, 26(3), 155–188. https://doi.org/10.1093/ijnp/pyac074
- Johnson, M. W., & Griffiths, R. R. (2017). Potential Therapeutic Effects of Psilocybin. Neurotherapeutics, 14(3), 734–740. https://doi.org/10.1007/s13311-017-0542-y
- U.S. Drug Enforcement Administration. (n.d.). Drug scheduling. https://www.dea.gov/drug-information/drug-scheduling.
- Johnson, M. W., Griffiths, R. R., Hendricks, P. S., & Henningfield, J. E. (2018). The abuse potential of medical psilocybin according to the 8 factors of the Controlled Substances Act. Neuropharmacology, 142, 143–166. https://doi.org/10.1016/j.neuropharm.2018.05.012
