Ruhee Mehta
Of the many diseases and conditions in the world, disability is one that can strike anyone at any moment. There are a number of ways it can occur: strokes or injuries to the spinal cord inducing paralysis, a horrible accident causing someone to lose a limb, and so many more. It’s a startling, profoundly disorienting and painful change to lose the ability to do something that you previously could. However, depending on the type of disability, prosthetics may be able to restore some of the lost function. If you lose your arm, maybe you can’t paint with your prosthetic hand but the prosthetic might enable you to pick things up. If you lose the ability to walk, maybe you won’t climb a mountain but you can climb stairs with prosthetic legs. With time, prosthetics have become better and people using them are able to do more and more things that they couldn’t before. Even so, there are limitations to what prosthetics can do and what forms of disability they help with.
Prosthetics can help restore function to varying degrees depending on the type of disability. For example, functions once performed by a lost limb can be partially substituted with the help of a prosthetic limb. Losing a limb is like the phone at the end of the line breaking; a prosthetic is the new, less ideal phone replacing it, so while it’s not perfect the telephone can still function to a degree. However, many conditions cannot be treated using a basic prosthetic. An example of a condition like that is paralysis induced by injury to the spinal cord. Much like a broken telephone wire that doesn’t permit phone calls to go through, a paralyzed person lacks the continuous connections between the neurons in the spinal cord that communicate commands from the brain to the limb. For people with spinal cord injuries, without intact telephone lines, the prosthetic limb cannot work. But there is hope: research in a new field of technology shows potential to restore movement to paralyzed people. This technology is called a ‘Brain-Computer Interface’ (BCI), also known as a ‘Brain-Machine Interface’ (BMI). Instead of requiring the telephone wires within the body to remain whole and capable of communicating commands, the BCI interfaces directly with the source: the brain. The technology uses instruments capable of deciphering commands directly from the brain, translates it into something a prosthetic limb can understand, and sends the command to the prosthetic limb along external wiring to perform the action (1).

Image of a woman wearing an EEG cap as a part of a BCI to operate a robotic arm. From Brain BCI, by Adindva1, 2017, Wikimedia Commons. https://commons.wikimedia.org/wiki/File:Brain_BCI.jpg CC BY-SA 4.0
It sounds fantastical – how are commands from the brain so easily picked up? How can thoughts and desires be comprehended by a machine? The answer begins by understanding how thoughts and actions are made physical within the brain. When thoughts or the desire to perform an action occur, the cells of the brain – better known as neurons – activate in concert, transmitting commands within the brain and to the body using self-produced electrical currents. Specific neurons in different areas of the brain activate in precise patterns depending on the type of action – whether it’s speaking, walking, or raising a hand to wave to a friend. The simpler the movement, the easier the pattern is to visualize and understand. For example, walking is far simpler than dancing. Even so, the simplest of actions are immensely complex. When you walk, you move your feet one after the other, but you also control your balance, make microadjustments to your gait, coordinate your toes to your heels, ankles, and other parts of your legs and conduct many other actions. This is all accounted for within your brain and by your neurons, mostly unconsciously. Consequently, to restore even simple function, a prosthetic must be able to pick up and interpret these complex commands from the brain (1).
How does the BCI, quite literally, read a person’s mind to understand what action they want to conduct? A number of different methods and techniques measure the changes in the brain’s electrical activity that signify movement and where in the brain that occurs. These methods are extremely invasive, requiring entry into the brain itself past the skull, or noninvasive, operating on top of the head. Devices such as electrodes, which are small metal conductors that pick up on the electrical activity within your brain, are highly invasive because they either touch the surface of the brain or are implanted within the brain. The invasiveness permits better detection and greater clarity of signal, as the signal is not interrupted by brain tissue and the electrode is placed at very specific areas on the brain to capture the signal from that region alone (2). However, having pieces of metal within your brain is not ideal for a person trying to go about their day, and is mostly utilized for research purposes. There are noninvasive devices, such as the electroencephalogram (EEG), which uses small metal discs attached to wires on the scalp to detect the changes in activity in the brain. While preferable, the clarity of signal is far lower due to the presence of brain tissue and the skull blocking it, as well as an inability to zero in on a hyper specific location of the brain. Consequently the prosthetic can interpret and perform far fewer movements with these noninvasive detectors compared to using invasive devices such as electrodes (3, 4).
As the change in electrical activity from the brain that command a limb to move is not instantly comprehensible to a machine, the signal must undergo processing to be ‘translated’. An essential part of the prosthetic, therefore, is the computer that analyzes and transforms that signal. Much like how a telephone book lists people’s names next to their phone numbers, the computer has a giant dataset that lists specific patterns of activity in the brain to a corresponding movement. That dataset was collected by analyzing thousands of people, and how their neuronal activity changes when they are about to perform specific actions. The way the computer analyzes and breaks down the signal to locate its position in the dataset is like how you might try to find someone’s name from their phone number in a telephone book. First, you look at their area code. After flipping to the part of the book that contains that code, you then look at the next three numbers, the local area identifier, to narrow down which section it might be in. And once you have narrowed down that section, you might go through the numbers individually until you find the correct phone number in the book and can then find that person’s name. The signal acquired from the brain similarly goes through an algorithm that breaks it down to understand what activity is correlated with. Once that signal is correlated to the appropriate movement within the dataset, the command is sent to the prosthetic to perform the movement (2).
The prosthetic itself is either a simple robotic limb that purely follows the mental commands sent by its user, or a ‘smart’ prosthetic. A smart prosthetic is capable of correcting tiny errors in movements without needing input from the brain – for example, adjusting the unconscious balance of a person walking to make sure they don’t fall. While robotic arms are used far more than robotic legs, brain-controlled wheelchairs are also being researched (4). This is especially revolutionary in the case for people who have spinal cord injuries that prevent them from being able to use a standard prosthetic limb. Using this technology gives people the ability to perform activities of daily living without being dependent on other people to do it for them.
Spinal cord injuries are especially well-suited for BCI use because of the nature of the damage in the condition. With spinal cord injuries the brain is fine, but the neurons – the aforementioned telephone wires – sending commands via the spinal cord to move the limbs are damaged. As BCIs bypass those spinal pathways and directly contact the brain, they restore a degree of function for people with this condition. The motor cortex is the region of the brain whose activity is detected by BCIs for movement-enabling prosthetics. It is responsible for controlling movement – it rehearses movements, plans movements, and makes the decision to perform the movement. The specific type of movement the motor cortex controls is voluntary movement, which is consciously controlled, unlike involuntary movements such as beating of the heart, churning of the stomach, and so on. A section of the motor cortex, called the primary motor cortex, sends out the command to perform the desired movement. The body knows what part to move because the human body is mapped out onto the primary motor cortex, and the section of the motor cortex activated corresponds with the appendage to be moved. The decision on the force, speed and direction with which the body part moves is made by a collection of neurons, that collectively ‘vote’ on their preferred direction. So when the detectors from the BCI touch or are in proximity to the primary motor cortex they are able to gather all the needed information on how the user wants the prosthetic to move. However, there is an issue: people are unique and so have tiny individual differences in how they activate their brain to move. While the dataset that BCIs use to correlate the thought pattern is large, it cannot account for each individual difference. Correspondingly, BCIs requires people to learn to think in specific ways that is conducive to the technology (1, 2). This occurs mostly through practice and feedback. The user repeatedly tries to think through their movements and plan them to move the prosthetic until they figure out what sort of sequence works best. For example, to grab a glass of water, the person operating the BCI must think through each step of reaching out, positioning their robotic hand, opening their fingers, closing them around the cup, and moving it in a straight line to the person. They may think about performing the motions in different ways until they figure out what way of thinking correlates best with the way they want to move. It requires conscious awareness of performing actions that are done in an able person almost without thought. This essentially rewires the sequence of activation in the brain. In order to enhance and accelerate the change in activation pattern to improve the user’s proficiency, virtual reality (VR) is frequently utilized before actual implementation of the BCI. After learning to control the prosthetic in VR, it is easier for them to work with the BCI in real life. This is especially vital as despite recent breakthroughs in usage of EEGs to control robotic limbs and wheelchairs, most clinical experiments with paralyzed people have only used implanted electrodes successfully (4).
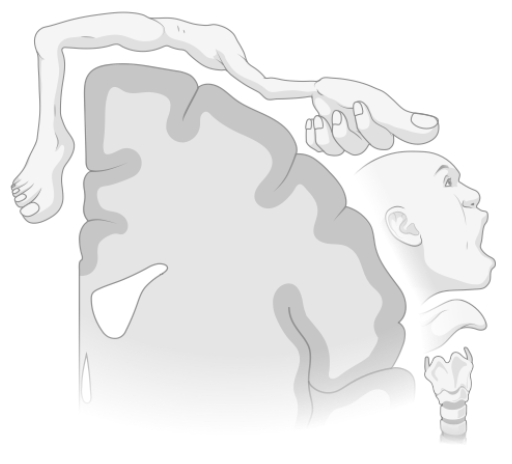
Image of the parts of the body mapped onto the primary motor cortex in the brain. Image from BioRender
Despite the immense promise that BCIs have for the world of prosthetics, there are many challenges faced before they can be used regularly for clinical purposes. The first lies in the detectors that perceive signals from the brain. The noninvasive ones are unable to achieve the same precision as the invasive ones, but the invasive ones work only from a few months to a year at a time. One of the reasons why is that the brain sometimes has an immune response to the foreign electrode, and activates its immune cells to surround the electrode in an attempt to prevent it from getting into contact with more tissue. This causes insulation of the electrode, and it cannot pick up signals as proficiently. The duration of efficacy of the electrodes is short given the riskiness of the surgery to implant them, as it requires opening up the skull to access the brain and so carries a risk of deadly infection. Another challenge is even after getting the invasive surgery, the electrodes are not always totally reliable in picking up signals. This partly occurs because of post-surgical scar tissue that interrupts the signal from the neurons, which is formed within the brain due to the friction from the electrodes. Currently, however, there is no verified method to reduce the scar tissue. A third problem is that implantation of invasive devices in the brain can disturb and damage other regions of the brain, including the frontal lobe. The frontal lobe is the seat of cognition and self-identity, and damage to it can lead to unpredictable and unwanted changes in sense of self. Is it worth risking losing who you are to regain your ability to move? Mistakes are costly, and long-term effects of having implanted electrodes are unknown, as this area is insufficiently researched. A fourth concern lies in challenges with the ability to correctly encode certain aspects of movements, specifically the trajectory and force used by the prosthetic to perform the desired movement. Encoding it is hard, complex, and researchers still have a long way to go before a lot of activities can be performed using the prosthetic (5, 6).
While BCIs show so much potential to help people, they also pose a number of ethical concerns around privacy and health equity. Ensuring any information gathered by a BCI remains confidential and cannot be hacked into is a major concern. In addition, BCIs are also extremely expensive, and require frequent calibration, check-ups, tuning, and so forth, which adds up quickly. It is unlikely to be easily available to the general public, or covered by all insurances. Consequently it unfortunately may become a device primarily used by the rich, or by less well-off people who are desperate and willing to conduct clinical trials (7).
In spite of the many challenges and obstacles that must be overcome to use BCIs, they are a life-changing technology with immense potential. While years of clinical trials and further research is required before BCIs could ever potentially be used broadly, they are worth studying and pursuing. They are capable not only of acting as a motor prosthetic for spinal cord injuries, but also as helpful tools for those who have had strokes that paralyzed a part of their body to rehabilitate effectively and regain use of that part. BCIs can even translate sounds from the machine to the brain, so those who are deaf can perceive sound. It includes helping people with disorders like Amyotrophic Lateral Sclerosis (ALS), who have lost the ability to speak and the coordination to type so they can communicate with the outside world again (8).
Breakthroughs in use and implementation of BCI technology are occurring every day. Recently, a team of scientists successfully used robotic surgery to implant electrodes in the motor cortex, enabling a paralyzed man to operate a robotic arm while maintaining the recording ability of the electrodes for over two hundred days (9). Accessibility is being improved slowly by scientists pioneering a Tele-BCI treatment for people who have had strokes that can be mostly done virtually, so people do not have to constantly come to the hospital or laboratory (10). New non-invasive neuron pattern detecting devices are being studied constantly to improve their resolution, pioneering a safer and easier way to utilize BCI prosthetics (5). These breakthroughs indicate the future of BCIs: prosthetics and rehabilitative technology that can help people like never seen before.
Literature Cited
- Zhang, H., Jiao, L., Yang, S., Li, H., Jiang, X., Feng, J., Zou, S., Xu, Q., Gu, J., Wang, X., & Wei, B. (2024). Brain-computer interfaces: the innovative key to unlocking neurological conditions. International Journal of Surgery (London, England), 110(9), 5745–5762. https://doi.org/10.1097/JS9.0000000000002022
- Batista A. (2020). Brain-computer interfaces for basic neuroscience. Handbook of Clinical Neurology, 168, 233–247. https://doi.org/10.1016/B978-0-444-63934-9.00017-2
- Edelman, B. J., Zhang, S., Schalk, G., Brunner, P., Muller-Putz, G., Guan, C., & He, B. (2025). Non-Invasive Brain-Computer Interfaces: State of the Art and Trends. IEEE Reviews in Biomedical Engineering, 18, 26–49. https://doi.org/10.1109/RBME.2024.3449790
- Huang, Q., Zhang, Z., Yu, T., He, S., & Li, Y. (2019). An EEG-/EOG-Based Hybrid Brain-Computer Interface: Application on Controlling an Integrated Wheelchair Robotic Arm System. Frontiers in Neuroscience, 13, 1243. https://doi.org/10.3389/fnins.2019.01243
- Bensmaia, S. J., & Miller, L. E. (2014). Restoring sensorimotor function through intracortical interfaces: progress and looming challenges. Nature Reviews. Neuroscience, 15(5), 313–325. https://doi.org/10.1038/nrn3724
- Maiseli, B., Abdalla, A. T., Massawe, L. V., Mbise, M., Mkocha, K., Nassor, N. A., Ismail, M., Michael, J., & Kimambo, S. (2023). Brain-computer interface: trend, challenges, and threats. Brain Informatics, 10(1), 20. https://doi.org/10.1186/s40708-023-00199-3
- Davidoff E. J. (2020). Agency and Accountability: Ethical Considerations for Brain-Computer Interfaces. The Rutgers Journal of Bioethics, 11, 9–20.
- Willett, F. R., Kunz, E. M., Fan, C., Avansino, D. T., Wilson, G. H., Choi, E. Y., Kamdar, F., Glasser, M. F., Hochberg, L. R., Druckmann, S., Shenoy, K. V., & Henderson, J. M. (2023). A high-performance speech neuroprosthesis. Nature, 620(7976), 1031–1036. https://doi.org/10.1038/s41586-023-06377-x
- Ikegaya, N., Mallela, A. N., Warnke, P. C., Kunigk, N. G., Liu, F., Schone, H. R., Verbaarschot, C., Hatsopoulos, N. G., Downey, J. E., Boninger, M. L., Gaunt, R., Collinger, J. L., & Gonzalez-Martinez, J. A. (2024). A novel robot-assisted method for implanting intracortical sensorimotor devices for brain-computer interface studies: principles, surgical techniques, and challenges. Journal of Neurosurgery, 142(5), 1280–1288. https://doi.org/10.3171/2024.7.JNS241296
- Mansour, S., Giles, J., Nair, K. P. S., Marshall, R., Ali, A., & Arvaneh, M. (2025). A clinical trial evaluating feasibility and acceptability of a brain-computer interface for telerehabilitation in patients with stroke. Journal of Neuroengineering and Rehabilitation, 22(1), 91. https://doi.org/10.1186/s12984-025-01607-x