Ketamine and It’s Applications
Ketamine, which is also referred to as Special K and KitKat, is most commonly known as an addictive, popular party drug. It is often illegally consumed by young adults at nightclubs, raves, and private parties, and can be misused in these environments to facilitate sexual assault. Despite this negative connotation, Ketamine has long held its legal status in the medical field. Since the 1970s, it has been approved for short-term sedation and anesthesia. Then, more recently, in 2019, it received FDA approval to legally combat treatment-resistant depression, a debilitating condition that affects about 350 million people worldwide [1]. The creation of novel depression treatments is critical, as first-line medications only alleviate symptoms in 50% of users, require between 3-4 weeks to take action, and can induce serious side effects [2].
The Consequences of the Media’s Unethical Promotion of Ketamine Therapy
Unsurprisingly, with this new application comes media attention. Series like the Secret Lives of Mormon Wives’ showcase clips of their characters undergoing Ketamine Therapy. Specifically, Jen and Zac Aflac use Ketamine on the show to “reset past traumas and habits.” Jen describes Zac trying Ketamine therapy as a representation of him “showing up for [their].. relationship” [3]. Cast member Demi Engemann has also spoken openly about her experience with Ketamine. According to US Weekly, she refers to Ketamine therapy as her “biggest tool” to strengthen her marriage; she says it will “spice up your sex life” and promote deep transformative conversations. As a result, she pleads for couples to “go do Ketamine” if their relationship is struggling or stagnant. Despite this clear endorsement from the show, the cast members fail to convey the drawbacks of Ketamine treatment. In fact, Demi refers to Ketamine as the “safest form of anesthesia” that can be used on “kids and small animals”. This same one-sided portrayal is shared by other public figures such as Elon Musk, who called it the “better option” for psychiatric depression treatment, and Lamar Odom, who used it to “feel amazing… [be] alive,” and stay sober [4].
Since celebrity opinions can significantly shape public perceptions, this one-sided portrayal of Ketamine therapy can have a dangerous influence on health-related decision-making. When endorsing products, celebrities can “catalyze herd behavior” away from competing options; in this context, a celebrity referral may cause an individual to choose Ketamine therapy over other antidepressant treatments without properly weighing the associated risks and benefits [5]. Neuroscience supports this behavioral response. When individuals view celebrity endorsements, they experience a rise in brain activity compared to when they are exposed to advertisements without famous endorsers [6]. This activity occurs in a brain region involved in forming positive associations between neutral and rewarding stimuli; its heightened activation suggests that audiences may implicitly link the endorsed product with the celebrity figure. Brain regions associated with memory formation also show increased activation; therefore, the individual may explicitly retrieve and connect past concepts of the public figure with the product. As a result, the audience’s opinion of the celebrity – whether positive or negative – can largely influence both their perception of the product and their willingness to engage with it [5, 6].
Providers offering Ketamine services or products may take advantage of these associations by using well-liked celebrities in their marketing. For example, on the Richmond Brain and Behavior website, there is a blog post with “heartfelt testimonials from well-known personalities who have found solace and relief through ketamine therapy”. The post offers eight ketamine-endorsing quotes from various well-known celebrities – ranging from Michael Phelps to Ryan Reynolds. These statements, while disclosing that Ketamine is not a “one-size-fits-all solution,” describe the drug as a “vital tool,” “game-changer,” and “life-line” [7]. What’s particularly concerning about these quotes is that they do not appear to be verifiable in any widely documented interview, article, or public statement. Since this company offers Ketamine treatment, this raises ethical concerns about the accuracy and intent of such messaging. By providing unverified celebrity testimonials without any scientific evidence to support their claims, it appears that the company may be using celebrities as a persuasive marketing technique.
However, even if the statement is coming directly from the public figure, it still must be evaluated critically. There is a common marketing technique on social media where influencers are paid large sums of money to endorse products they may have never actually used. These posts are made to promote a wide range of product-types including cosmetics, beverages, vitamins, and more. Research suggests that over 60% of consumers trust these online recommendations [8]. While spreading misinformation is never okay, the risk of purchasing may be small for non-health-related or well-researched items. However, these uninformed advertisements are extremely dangerous for promoting drugs like ketamine that lack clear long-term research and carry significant side effects. These can lead to inadequate and misinformed healthcare, having lasting mental and physical consequences for the viewer. Therefore, it is imperative that individuals are fully educated on the pros and cons of Ketamine treatment so that they can critically evaluate and not fall victim to these one-sided celebrity endorsements. This article aims to provide the audience with the resources and knowledge necessary to make these informed decisions on their own.
How does Ketamine Work as an Antidepressant?
Ketamine therapy functions as a treatment for depression by exerting an effect on the basic building blocks of the brain, called neurons. These specialized brain cells send and receive information, allowing the brain to effectively regulate mood, behavior, and diverse bodily functions. While neurons have various components that support their function, there are two main structures important for communication: (1) the dendrites, which receive incoming signals from other neurons and (2) the axon terminals, which send signals onward to the next neuron.
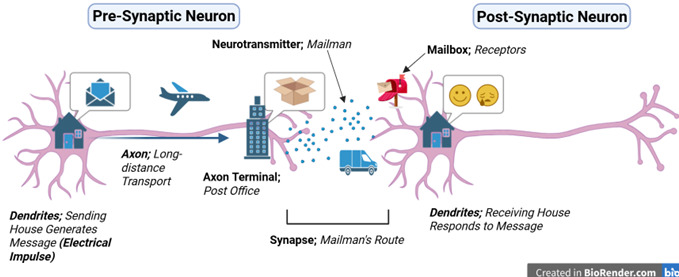
Communication between neurons can be understood by referencing a mail system. This delivery process begins when an electrical impulse – or message – is generated in the dendrite of the first neuron, known as the pre-synaptic neuron. This message travels down the neuron’s axon, similar to how mail is transported by trucks and planes between distant processing centers. Once this message reaches the local post office – or axon terminal – it is then prepared for delivery by being packaged into a chemical messenger molecule, called a neurotransmitter. Neurotransmitters deliver a message from one neuron to the next, just like a mailman transports letters. To do so, these molecules of information travel across a small gap between neurons called the synapse, which represents the mailman’s route from the post office to the receiving house. When the neurotransmitter reaches the house, it arrives at the dendrites of the second, post-synaptic cell. There, it binds to a specific receptor protein, or mailbox, that only accepts mail of certain shapes and sizes. When the message arrives at the correct mailbox, it opens the mailbox, triggering a response in the receiving neuron, just like humans respond to the content of a letter. This response influences the type of message that neuron will pass along; messages can be either excitatory – promoting more delivery – or inhibitory – slowing down the mail system [9].
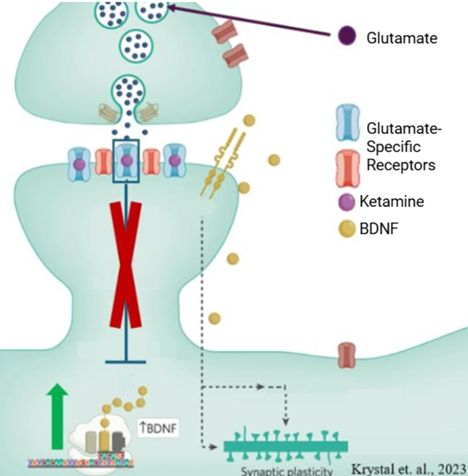
Figure 3: Ketamine blocks glutamate-specialized receptors. This increases expression of BDNF to subsequentially treat depression symptoms. Image adapted from [10]
Ketamine works by mediating the action of a particular messenger called glutamate. Glutamate is known as the brain’s main excitatory neurotransmitter; it binds to glutamate-specialized receptors on the postsynaptic neuron to promote further neuronal communication. In the absence of ketamine, glutamate attachment activates the receptor, which in-return initiates different sequences of events called pathways. One of those pathways decreases levels of a molecule known as Brain Derived Neuronal Growth Factor (BDNF). BDNF acts as a fertilizer for neurons, promoting neuronal growth and survival. However, when Ketamine is present in the synapse, it blocks the action of glutamate’s receptor. This stops the pathway that reduces BDNF expression. Therefore, levels of BDNF rise, and neurons can grow and adapt. Ketamine, as a result, works quickly to alleviate depression symptoms by supporting the repair of malfunctioning neural connections that are related to mood regulation [10].
How Are the Side Effects of Ketamine Therapy Assessed?
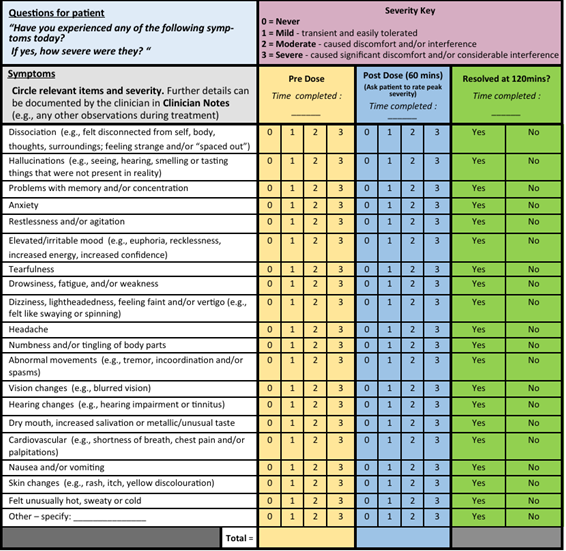
Figure 4: Example questions from the acute treatment module of the Ketamine Side Effect Tool (KSET). Similar questions are used during the baseline and follow-up modules; however, symptom severity is only assessed at one time point. Image taken from [2].
The Ketamine Side Effect Tool (KSET) was developed to assess the health consequences and safety of ketamine treatment. The test consists of four distinct modules – screening, baseline, acute treatment, and follow-up. The screening section documents any medical history that could affect the individual’s response to Ketamine, such as cardiovascular issues, prior substance abuse, and neurological disorders. The remaining three modules use a series of questions to evaluate the patient’s symptom severity overtime; all questions are answered on a scale from 0 (never) to 3 (severe). The baseline and follow-up questionaries are identical, and both contain memory tests to evaluate ketamine’s impact on cognition. Conversely, while similar, the acute treatment module provides a more comprehensive assessment by asking patients to rate their symptoms at multiple timepoints following the administered dose. By comparing these measurements to baseline and follow-up values, the KSET allows for evaluation of both acute and long-term treatment consequences [2, 17].
The Success of Ketamine Therapy
For patients that are resistant to typical depression interventions – such as Zoloft, Lexapro, Prozac, and Wellbutrin – ketamine therapy can rapidly alleviate symptoms, sometimes in in less than two hours [11]. Treatments are typically administered as a series of 4 to 6 infusions over a 2-to-3-week period [12]. Research shows that for single doses of ketamine therapy, 77% of patients respond well to treatment and 43% of patients experience reduced symptoms [13]. In comparison, long-term treatment is associated with less sustained positive outcomes. After four doses of ketamine therapy, 26.7% of individuals were found to have reduced symptomology, while only 13.3% of patients still felt this positive effect after a twelve-month follow-up [12]. Ketamine treatment has also been shown to directly reduce depressive symptoms, improving scores on standardized depression scales by 37.8% [14]. It is also highly effective at reducing suicide ideation, with significant effects seen as soon as 40 minutes post-infusion [15]. In contrast to traditional treatments, ketamine is shown to produce significantly larger depression scale reductions, higher quality of life scores, and lower side effect reports. Additionally, patients are more likely to follow their treatment plan, with an adherence rate of 91% for ketamine therapy compared to 84% for first-line treatments such as Zoloft [16].
The Drawbacks and Potential Side Effects of Ketamine Therapy
Even though ketamine can effectively treat depression, its benefits may be relatively short-lived, with symptom relapses commonly reported. Even in studies demonstrating strong initial antidepressant effects, the benefits often wane by the end of the observation period. Some studies have found that the antidepressant response disappears within three to four days, while others report it diminishing after one to three weeks [13, 14]. However, there is evidence that modifying treatment parameters such as frequency, duration, and dosage may extend its therapeutic benefits; for instance, after seven months of weekly ketamine sessions, improvements in depressive symptoms persisted for an additional six months [13].
While ketamine therapy is beneficial for some patients, it carries a wide range of potential side effects that must be thoroughly considered before treatment. According to a large-scale analysis performed using the KSET, ketamine therapy is found to have extremely high rates of dissociative effects – a feeling of extreme disconnectedness from the mind or body. This was reported to occur in over 90% of patients and during 68.8% of appointments. Cognitive and psychological detriments are also frequent, with nearly half of patients reporting problems with memory and concentration, and over 20% experiencing hallucination-like sensory changes [17]. However, experts suggest that when used appropriately and at low doses, ketamine causes cognition to remain stable or improve overtime [18]. Other common psychiatric side-effects include anxiety, depression, panic attacks, confusion, and headaches [13,18]. It’s important to note that the risk for most of these side effects is only present during ketamine administration and subsides once treatment is over. Beyond the immediate treatment window, the most common inter-session symptoms are sleep disturbances, gastrointestinal issues, tremors, poor coordination, and irritable mood [2,17]. Ketamine therapy is also shown to moderately increase the risk of physical effects such as nausea, dizziness, urinary tract complications, liver toxicity, and reduced oxygen saturation [13]. 38% of studies also reported acute heart and circulatory related changes [18].
There are also concerns about the potential for drug abuse and dependence. While Ketamine does carry this risk, it’s less likely to occur for low-dose medical sessions than recreational use. In a study of patients undergoing ketamine treatment for 6 months, there was no evidence of ketamine craving or usage outside of the study context [19]. However, there is still some potential for misuse with treatment. For example, celebrity Matthew Perry passed away at age 54 from the “acute effects of ketamine.” While Perry was utilizing ketamine illegally at the time of his passing, he first started receiving regular infusions of the substance to treat severe depression and anxiety at a local clinic. His last legal infusion took place about a week before this death; this heartbreaking story highlights the need for close patient observation, especially for those at risk of addiction [4].
In Conclusion
The evidence in this article should be utilized as a resource to evaluate the potential risks and benefits of ketamine therapy. It is especially important to consider these empirical findings as celebrity endorsements or marketing-heavy advertisements are viewed. Companies and public figures should reconsider the ethicality of these promotions and should modify their marketing approach to include accurate testimonies with a full transparency of any potential side-effects. However, this change can’t be relied on, so audiences should conduct their own informed research before making any medical decisions.
References
- Department of Justice (2020). Ketamine drug fact sheet. https://www.dea.gov/sites/default/files/2020-06/Ketamine-2020_1.pdf
- Short, B., Fong, J., Galvez, V., Shelker, W., & Loo, C. K. (2018). Side-effects associated with ketamine use in depression: a systematic review. The Lancet Psychiatry, 5(1), 65–78. 10.1016/S2215-0366(17)30272-9
- Jenkins, J., Secret Lives of Mormon Wives (2025). Jeffery Jenkins Production Company
- US Weekly (2026). https://www.usmagazine.com/
- Hoffman, S. J., & Tan, C. (2015). Biological, psychological and social processes that explain celebrities’ influence on patients’ health-related behaviors. Archives of Public Health, 73(1). https://doi.org/10.1186/2049-3258-73-3
- Stallen, M., Smidts, A., Rijpkema, M., Smit, G., Klucharev, V., & Fernández, G. (2010). Celebrities and shoes on the female brain: The neural correlates of product evaluation in the context of fame. Journal of Economic Psychology, 31(5), 802–811. https://doi.org/10.1016/j.joep.2010.03.006
- Beasley, B. (2023, November 6). Celebrities Praise the Healing Power of Ketamine Therapy: Testimonials That Inspire. Richmond Brain and Behavior. https://www.richmondbrainandbehavior.com/post/celebrities-praise-the-healing-power-of-ketamine-therapy-testimonials-that-inspire-1
- Ekinci, Y., Dam, S., & Buckle, G. (2025). The Dark Side of Social Media Influencers: A Research Agenda for Analyzing Deceptive Practices and Regulatory Challenges. Psychology and Marketing, 42(4). https://doi.org/10.1002/mar.22173
- Ludwig, P. E., Reddy, V., & Varacallo, M. (2023, July 24). Neuroanatomy, Neurons. PubMed; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK441977/
- Krystal, J. H., Kavalali, E. T., & Monteggia, L. M. (2023). Ketamine and rapid antidepressant action: new treatments and novel synaptic signaling mechanisms. Neuropsychopharmacology, 49. https://doi.org/10.1038/s41386-023-01629-w
- Zarate, C. A., Singh, J. B., Carlson, P. J., Brutsche, N. E., Ameli, R., Luckenbaugh, D. A., Charney, D. S., & Manji, H. K. (2006). A Randomized Trial of an N-methyl-D-aspartate Antagonist in Treatment-Resistant Major Depression. Archives of General Psychiatry, 63(8), 856. https://doi.org/10.1001/archpsyc.63.8.856
- Sakurai, H., Daiki Setoyama, Kato, T. A., Hisateru Tachimori, Murao, M., Matsumoto, Y., Tada, T., Imamura, Y., Seki, H., Tsuboi, T., Uchida, H., & Watanabe, K. (2025). Acute and longterm effects of repeated ketamine infusions in treatment‐resistant depression and associated metabolite changes. Psychiatry and Clinical Neurosciences. https://doi.org/10.1111/pcn.13870
- Katalinic, N., Lai, R., Somogyi, A., Mitchell, P. B., Glue, P., & Loo, C. K. (2013). Ketamine as a new treatment for depression: A review of its efficacy and adverse effects. Australian & New Zealand Journal of Psychiatry, 47(8), 710–727. https://doi.org/10.1177/0004867413486842
- Wilkinson, S. T., Katz, R. B., Toprak, M., Webler, R., Ostroff, R. B., & Sanacora, G. (2018). Acute and Longer-Term Outcomes Using Ketamine as a Clinical Treatment at the Yale Psychiatric Hospital. The Journal of Clinical Psychiatry, 79(4). https://doi.org/10.4088/jcp.17m11731
- Ballard, E. D., Ionescu, D. F., Vande Voort, J. L., Niciu, M. J., Richards, E. M., Luckenbaugh, D. A., Brutsché, N. E., Ameli, R., Furey, M. L., & Zarate, C. A. (2014). Improvement in suicidal ideation after ketamine infusion: Relationship to reductions in depression and anxiety. Journal of Psychiatric Research, 58, 161–166. https://doi.org/10.1016/j.jpsychires.2014.07.027
- Nawaz, A., Mamoon, B., Batool, T. E., Khattak, M. I., Amir, F., Akbar, A., & Khan, S. (2024). Advances in Antidepressant Therapy: Comparing the Efficacy of Selective Serotonin Reuptake Inhibitors (SSRIs), Serotonin-Norepinephrine Reuptake Inhibitors (SNRIs), and Novel Agents. Cureus, 16(12). https://doi.org/10.7759/cureus.76318
- Bayes, A., Cao, T. V., Barreiros, A. R., Massaneda-Tuneu, C., Dong, V., Thornton, N., Glozier, N., Beesley, L., Moreno, D., Gálvez-Ortiz, V., Short, B., Martin, D., & Loo, C. (2024). Safety outcomes of ketamine for treatment-resistant depression in clinical settings and development of the ketamine side effect tool-revised (KSET-R). Psychiatry Research, 344, 116334. https://doi.org/10.1016/j.psychres.2024.116334
- Nikayin, S., Murphy, E., Krystal, J. H., & Wilkinson, S. T. (2022). Long-term safety of ketamine and esketamine in treatment of depression. Expert Opinion on Drug Safety, 21(6), 1–11. https://doi.org/10.1080/14740338.2022.2066651
- Perry, E. B., Cramer, J. A., Cho, H.-S., Petrakis, I. L., Karper, L. P., Genovese, A., O’Donnell, E., Krystal, J. H., & D’Souza, D. C. (2007). Psychiatric safety of ketamine in psychopharmacology research. Psychopharmacology, 192(2), 253–260. https://doi.org/10.1007/s00213-007-0706-2