It is likely not a stretch to say that most people have had a grandparent who has experienced issues related to aging. This can be anything from memory problems to keeping in number two. One of the most prominent aging-related diseases is Alzheimer’s disease. In fact, aging is the single greatest risk factor for Alzheimer’s disease (1). Individuals between the age of 75 and 84 have an estimated 19% increase in chance for Alzheimer’s disease development, while this estimate blooms to 30-35% in those older than 85 years of age (2,3). Other than the horrible experience of seeing a loved one’s cognitive function deteriorate, Alzheimer’s disease also presents a large economic burden. The global cost of dementia (of which Alzheimer’s disease is a version of) is expected to reach 2 trillion dollars by 2030 (4). Whether one has experience with someone who has Alzheimer’s disease or not, there is clearly a reason to be concerned.
Aging is a process that every person goes through and therefore all people have a certain risk for developing Alzheimer’s disease. But what if I told you that regardless of aging, some populations have a greater risk for Alzheimer’s disease. Income, occupation, and education are three factors that contribute to one’s risk for the development of Alzheimer’s disease. Together, these three factors make up socioeconomic status. Populations with low socioeconomic status have an increased risk for the development of Alzheimer’s disease. Many people are unaware, but there is a large disparity in longevity and aging depending on the socioeconomic bracket that individuals are in. It is necessary to spread awareness about this fact and to understand how low socioeconomic status predisposes a person to Alzheimer’s disease.
The socioeconomic status of a person is the relative position they have within the societal hierarchy. People with low incomes, less occupational prestige, and lower levels of educational attainment are considered to have less socioeconomic status than people with more income, occupational prestige and educational attainment. A study of 344,324 participants across a 15-year period that looked at 210 modifiable risk factors predicted that in 13.5% of cases, having a higher socioeconomic status could have contributed to the prevention of dementia development (5). Another study with nearly 1.5 million individuals found that people with less educational attainment and income had an increased risk for cognitive impairment and developing dementia (6). This was replicated in another 2023 study that looked at education as a protective factor for Alzheimer’s disease. Researchers used genetics to predict how many years of schooling a person would take part in. They found that for about every 4 years of predicted schooling, a person’s chance to develop Alzheimer’s disease was decreased by 30% (7). But why is lower socioeconomic status associated with an increased risk for Alzheimer’s disease? The connection seems to be through inflammation in the body and brain.
Inflammation is the body’s response to injury or insult. The immune system releases chemicals in response to injury or insult, which can amplify (pro-inflammatory) or reduce (anti-inflammatory) inflammation. These chemicals are generally known as cytokines. Pro-inflammatory cytokines turn up the stress response, so if inflammation takes place over a long period of time, then a person has an increased risk of being chronically stressed. Inflammation often gets a bad rap because it can be linked to almost every acute or chronic disease that a person can experience. Acute inflammation is part of the body’s healing process and only becomes negative when inflammation continues at low levels for a long period of time, which is called chronic inflammation. Chronic inflammation is what contributes to chronic disease.
Socioeconomic stressors such as poverty, job security, and unemployment upregulate chronic inflammatory processes. The release of pro-inflammatory cytokines disrupts processes affected by Alzheimer’s disease such as processing speed, memory speed, and learning (8). Pro-inflammatory cytokines play a role in the production of two measurable indicators of Alzheimer’s disease: tau tangles and amyloid beta plaques. Measurable indicators of a biological state or condition are called biomarkers. Generally, the presence of tau tangles and amyloid beta plaques indicate a person has Alzheimer’s disease. The amount of tau tangles is thought to be correlated with the level of cognitive impairment in people with Alzheimer’s disease, while amyloid beta plaques break down the brain (9).
One way to measure inflammation is through the presence of C-reactive protein (CRP) (10). CRP is responsible for helping to identify what is damaging the body, and aids in getting rid of debris. In most cases however, CRP is thought of as a biomarker of inflammation. CRP is synthesized in the liver in response to immune system activation, then sent to the blood stream where it is found at its highest concentrations (11).
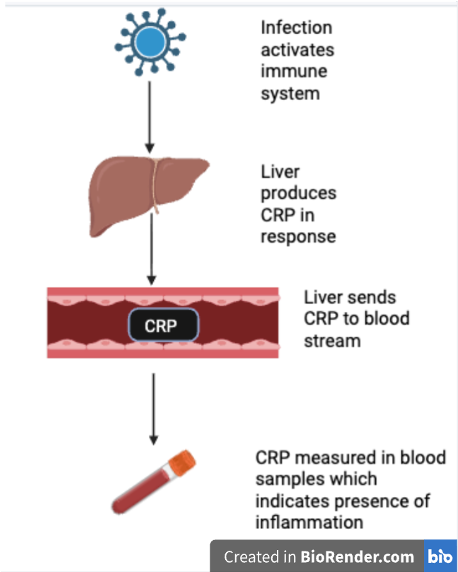
An individual’s CRP concentration can be measured by determining the amount of CRP per liter of blood present in a blood sample. Blood samples are taken through something called an ELISA assay. In an ELISA assay, an antibody binds to a target protein, which allows the target protein to be quantified (12). The antibody is like a key that can only unlock a specific lock, the antigen. In this case, an antibody will bind to an antigen specific to CRP, which allows for the quantification of CRP. The average CRP concentration is about 0.8 milligrams per liter (11). CRP concentrations above 3 milligrams per liter, but less than 10 milligrams per liter are considered indicative of chronic inflammation. Ten milligrams per liter is considered representative of acute inflammation as opposed to chronic inflammation (10). It is important to note that CRP is not analogous to inflammation and does not cause inflammation. The relationship between CRP and inflammation can be understood through the metaphor of a burning building. In this hypothetical burning building, the fire can be considered inflammation because it does actual damage to the structure, CRP is smoke because where this smoke there is fire, just as when there is CRP there is inflammation. This all happens inside a building which is the human body in this case. In other words, inflammation (fire) signals the liver to begin producing CRP (smoke). Therefore, the production of CRP (smoke) is evidence that inflammation (fire) is present in the body (building).
CRP is particularly relevant to our topic because it has been utilized as an indicator of inflammatory states associated with socioeconomic stressors (13). Individuals with a lower socioeconomic status have higher levels of CRP at mid and later life compared to individuals from a higher socioeconomic status (14,15). Participants from the least advantaged families in childhood have 25% higher CRP levels than those from the most advantaged families (8). Additionally, unemployed people have significantly higher levels of inflammation than those who are employed (10). High CRP levels at midlife increase the risk for slight cognitive decline in participants without dementia, which is a diagnoseable condition known as mild cognitive impairment (16,17). A study of over 3,000 Americans with an average age of 74 found that those with high CRP levels (defined as above 3.0 milligrams per liter of blood) had a 24% greater risk for cognitive decline than those with CRP levels below this threshold (18). Mild cognitive impairment is not Alzheimer’s disease, but still represents cognitive decline, which is an unwanted side effect of aging, or in this case, chronic inflammation as a result of elevated CRP levels due to low socioeconomic status.
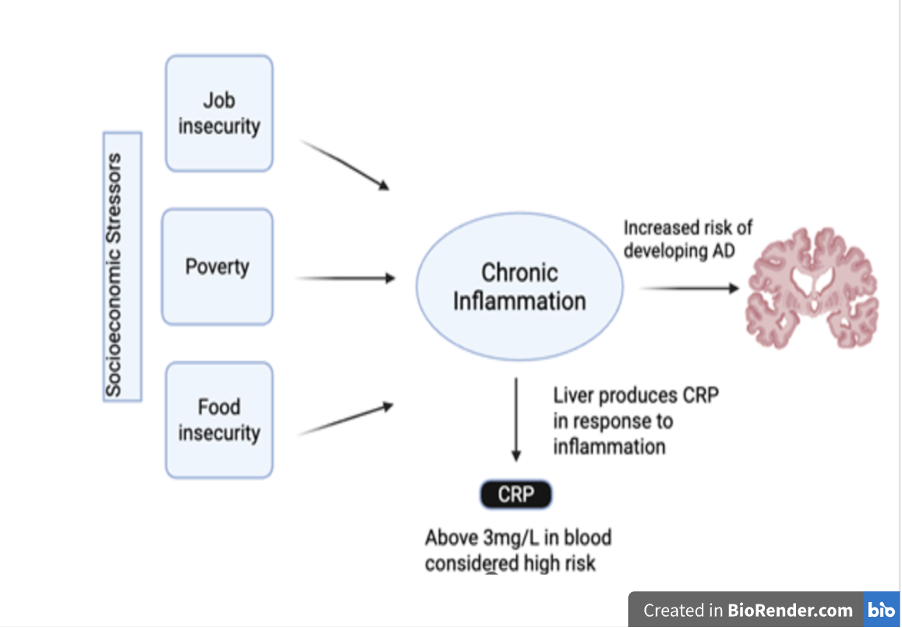
So now that we have talked about all the ways that inflammation, socioeconomic status and Alzheimer’s disease are related to each other, let’s put it all together. There is an association between socioeconomic status and Alzheimer’s disease risk that is partially dependent on an individual’s concentration of CRP. Genes that predispose a person to more years of education and higher household income are causally related to a decreased risk of developing Alzheimer’s disease. Additionally, higher educational attainment is associated with a reduction in CRP levels, which decreases the risk of developing Alzheimer’s disease. However, when individuals with a predisposition to more educational years have elevated CRP levels, they were no longer protected against Alzheimer’s disease. Specifically, about half of the total positive effect that education has on the decreased risk for Alzheimer’s disease can be offset by high CRP levels (19). This is unfair, especially for low socioeconomic status groups who are at risk for chronic inflammation and chronic stress.

The purpose of this article is twofold. I first wanted to inform you about the relationship between socioeconomic status, inflammation, and Alzheimer’s disease risk. We have learned that individuals with low socioeconomic status are more likely to develop Alzheimer’s disease than those with higher socioeconomic status. We also learned that socioeconomic stressors contribute to chronic stress and chronic inflammation, which are also related to Alzheimer’s disease risk. Second, we learned that elevated levels of CRP are indicative of chronic inflammation which mediates the protective relationship of educational attainment against the development of Alzheimer’s disease. It is without a doubt fascinating that inflammation can play such a large role in the development of chronic disease, however this is also extremely unfair.
There are times when socioeconomic status is determined by an individual, but for the most part people are born into a low, moderate, or high socioeconomic status. People cannot control what socioeconomic status they are born into which means they also cannot control the fact that they will have an increased chance for the development of a debilitating neurodegenerative disease. Consider the case of a person born into a low socioeconomic status, but that works extremely hard to overcome their situation and ends up having high educational attainment. Typically, this person would have education on their side as a strong protective factor against Alzheimer’s disease, but if this person still has socioeconomic stressors which increase their CRP levels indicative of chronic inflammation, then that protective effect will be mitigated. Fortunately, whether an individual is at a high or low risk for Alzheimer’s disease, they can engage in preemptive treatment practices such as exercise, mindfulness, and sleep. However, those born into a low socioeconomic status will likely still have a greater risk for Alzheimer’s disease then a person who does not have to deal with socioeconomic stressors and engages in preemptive practices.
So, how can you as a reader work to mitigate this unfair difference in Alzheimer’s disease risk? The first step is exposure, which you have done by reading this article. The next step would be to make more people aware of this public health discrepancy. A third step would be to research and advocate for policies that reduce socioeconomic disparities. There is a necessity for more epidemiological research so that public health experts can identify chronic disease trends. Community/population wide programs will likely be essential to mitigating the risk of Alzheimer’s disease in low socioeconomic status areas. These programs should be targeted at stopping generational patterns of Alzheimer’s disease risk, which will lead to a healthier future for all of society. Critics may cite the potential expense of these programs, but the quality of human life goes beyond monetary importance, and it is difficult to believe these programs would cost more than the trillions of dollars put into Alzheimer’s disease research. However, change can only be made if the public is aware of this issue and that starts with people like you spreading the message.
References
1. Hao, J. (2019). Risk Factors and Clinical Treatments of Alzheimer’s Disease. Journal of Physics: Conference Series, 1168, 032120. https://doi.org/10.1088/1742-6596/1168/3/032120
2. Ferri, C. P., Prince, M., Brayne, C., Brodaty, H., Fratiglioni, L., Ganguli, M., Hall, K., Hasegawa, K., Hendrie, H., Huang, Y., Jorm, A., Mathers, C., Menezes, P. R., Rimmer, E., & Scazufca, M. (2005). Global prevalence of dementia: A Delphi consensus study. The Lancet, 366(9503), 2112–2117. https://doi.org/10.1016/S0140-6736(05)67889-0
3. Knopman, D. S. (2001). An overview of common non-alzheimer dementias. Clinics in Geriatric Medicine, 17(2), 281–301. https://doi.org/10.1016/S0749-0690(05)70069-0
4. Tay, L. X., Ong, S. C., Tay, L. J., Ng, T., & Parumasivam, T. (2024). Economic Burden of Alzheimer’s Disease: A Systematic Review. Value in Health Regional Issues, 40, 1–12. https://doi.org/10.1016/j.vhri.2023.09.008
5. Zhang, Y., Chen, S.-D., Deng, Y.-T., You, J., He, X.-Y., Wu, X.-R., Wu, B.-S., Yang, L., Zhang, Y.-R., Kuo, K., Feng, J.-F., Cheng, W., Suckling, J., David Smith, A., & Yu, J.-T. (2023). Identifying modifiable factors and their joint effect on dementia risk in the UK Biobank. Nature Human Behaviour, 7(7), 1185–1195. https://doi.org/10.1038/s41562-023-01585-x
6. Wang, A.-Y., Hu, H.-Y., Ou, Y.-N., Wang, Z.-T., Ma, Y.-H., Tan, L., & Yu, J.-T. (2023). Socioeconomic Status and Risks of Cognitive Impairment and Dementia: A Systematic Review and Meta-Analysis of 39 Prospective Studies. The Journal of Prevention of Alzheimer’s Disease, 10(1), 83–94. https://doi.org/10.14283/jpad.2022.81
7. Seyedsalehi, A., Warrier, V., Bethlehem, R. A. I., Perry, B. I., Burgess, S., & Murray, G. K. (2023). Educational attainment, structural brain reserve and Alzheimer’s disease: A Mendelian randomization analysis. Brain, 146(5), 2059–2074. https://doi.org/10.1093/brain/awac392
8. Liu, R. S., Aiello, A. E., Mensah, F. K., Gasser, C. E., Rueb, K., Cordell, B., Juonala, M., Wake, M., & Burgner, D. P. (2017). Socioeconomic status in childhood and C reactive protein in adulthood: A systematic review and meta-analysis. Journal of Epidemiology and Community Health, 71(8), 817–826. https://doi.org/10.1136/jech-2016-208646
9. SantaCruz, K., Lewis, J., Spires, T., Paulson, J., Kotilinek, L., Ingelsson, M., Guimaraes, A., DeTure, M., Ramsden, M., McGowan, E., Forster, C., Yue, M., Orne, J., Janus, C., Mariash, A., Kuskowski, M., Hyman, B., Hutton, M., & Ashe, K. H. (2005). Tau Suppression in a Neurodegenerative Mouse Model Improves Memory Function. Science, 309(5733), 476–481. https://doi.org/10.1126/science.1113694
10. Hughes, A., Kumari, M., McMunn, A., & Bartley, M. (2017). Unemployment and inflammatory markers in England, Wales and Scotland, 1998–2012: Meta-analysis of results from 12 studies. Brain, Behavior, and Immunity, 64, 91–102. https://doi.org/10.1016/j.bbi.2017.03.012
11. Sproston, N. R., & Ashworth, J. J. (2018). Role of C-Reactive Protein at Sites of Inflammation and Infection. Frontiers in Immunology, 9, 754. https://doi.org/10.3389/fimmu.2018.00754
12. Hayrapetyan, H., Tran, T., Tellez-Corrales, E., & Madiraju, C. (2023). Enzyme-Linked Immunosorbent Assay: Types and Applications. In R. S. Matson (Ed.), ELISA (Vol. 2612, pp. 1–17). Springer US. https://doi.org/10.1007/978-1-0716-2903-1_1
13. Juster, R.-P., McEwen, B. S., & Lupien, S. J. (2010). Allostatic load biomarkers of chronic stress and impact on health and cognition. Neuroscience & Biobehavioral Reviews, 35(1), 2–16. https://doi.org/10.1016/j.neubiorev.2009.10.002
14. Hughes, A., McMunn, A., Bartley, M., & Kumari, M. (2015). Elevated inflammatory biomarkers during unemployment: Modification by age and country in the UK. Journal of Epidemiology and Community Health, 69(7), 673–679. https://doi.org/10.1136/jech-2014-204404
15. Janicki-Deverts, D., Cohen, S., Matthews, K. A., & Cullen, M. R. (2008). History of Unemployment Predicts Future Elevations in C-Reactive Protein among Male Participants in the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Annals of Behavioral Medicine, 36(2), 176–185. https://doi.org/10.1007/s12160-008-9056-5
16. Fernandes, A., Tábuas-Pereira, M., Duro, D., Lima, M., Gens, H., Santiago, B., Durães, J., Almeida, M. R., Leitão, M. J., Baldeiras, I., & Santana, I. (2020). C-reactive protein as a predictor of mild cognitive impairment conversion into Alzheimer’s disease dementia. Experimental Gerontology, 138, 111004. https://doi.org/10.1016/j.exger.2020.111004
17. Roberts, R. O., Geda, Y. E., Knopman, D. S., Boeve, B. F., Christianson, T. J. H., Pankratz, V. S., Kullo, I. J., Tangalos, E. G., Ivnik, R. J., & Petersen, R. C. (2009). Association of C‐reactive protein with mild cognitive impairment. Alzheimer’s & Dementia, 5(5), 398–405. https://doi.org/10.1016/j.jalz.2009.01.025
18. Tang, J., Lu, Q., Lin, H., Li, J., Zhu, X., & Ma, T. (2025). C‐reactive protein‐mediated dementia. Psychogeriatrics, 25(3), e70032. https://doi.org/10.1111/psyg.70032
19. Geng, C., Meng, K., & Tang, Y. (2024). Identifying the mediating role of inflammation on the relationship between socioeconomic status and Alzheimer’s disease: A Mendelian randomization analysis and mediation analysis. Journal of Neurology, 271(5), 2484–2493. https://doi.org/10.1007/s00415-023-12176-1