The word “toxin” is often thrown around in many online spaces, especially by so-called “health influencers” who claim everything they can’t pronounce on an ingredients list is incredibly harmful to your health. However, it’s important to remember that any substance in high enough amounts can be toxic. Think of the fact that bananas are radioactive: you would need to consume 40 million bananas in order to die of radiation poisoning. Apart from the fact nobody in their right mind would do this, it’s all a matter of perspective. When one hears neurotoxin in any capacity, their minds may jump to any number of creatures from Australia known for subduing or killing their prey by using such tactics as neurotoxins. It is important to note, however, that neurotoxins aren’t inherently evil or bad. In fact, a number of them have been used to treat conditions throughout history. In this article, I plan to present several neurotoxins that have had their uses in clinical settings for treatment.
Acetylcholine and the Neuromuscular Junction
Before we go into the specifics of the neurotoxins, we must first break down the systems upon which the toxins mentioned in this article function. The main neurotransmitter we’re going to cover: acetylcholine. For starters, neurotransmitters are chemical signals sent from one brain cell, or neuron, to another neuron, a muscle cell, or gland. The components we’re going to discuss are called the neuromuscular junction (NMJ), composed of specialized neurons that help plan and execute motor movements, known as motor neurons, and the skeletal muscle fiber. Those motor neurons being in the spinal cord and travel anywhere where movement may be necessary in your body. Acetylcholine (ACh) is the primary neurotransmitter involved with motor function. One important factor to note is that acetylcholine is what we refer to as an excitatory neurotransmitter, meaning that it excites the nerve cell to fire a message. It is created by the synthesis of choline and acetyl coenzyme A (acetyl-CoA) by an enzyme called choline acetyltransferase (ChAT). Once it’s synthesized, it’s then packaged and stored (1, Khalil et al., 2025). Acetylcholine is primarily stored in the end of nerve cells until it’s ready to be released. So how does it get released, and what does it do?
It all begins with something called an action potential. An action potential (AP) is the electrical impulse that travels down the neuron to trigger it to release the neurotransmitters to whatever site it needs to reach. The neurotransmitters are put into packages called vesicles and moved down the synapse. The vesicle fuses with the site, releasing the ACh contained into the gap between the neuron and the binding site, called the synaptic cleft. Binding itself is highly specialized and specific to a particular neurotransmitter, hormone, drug, or signaling molecule. In a way, you could think of each receptor as a lock, where the molecule acts as a key to trigger the tumblers in just the right way to produce an action.
Figure 2
Illustration of the en:disc tumbler lock created by Wapcaplet in Blender and touched up in the en:GIMP. en:Category:Images created with Blender
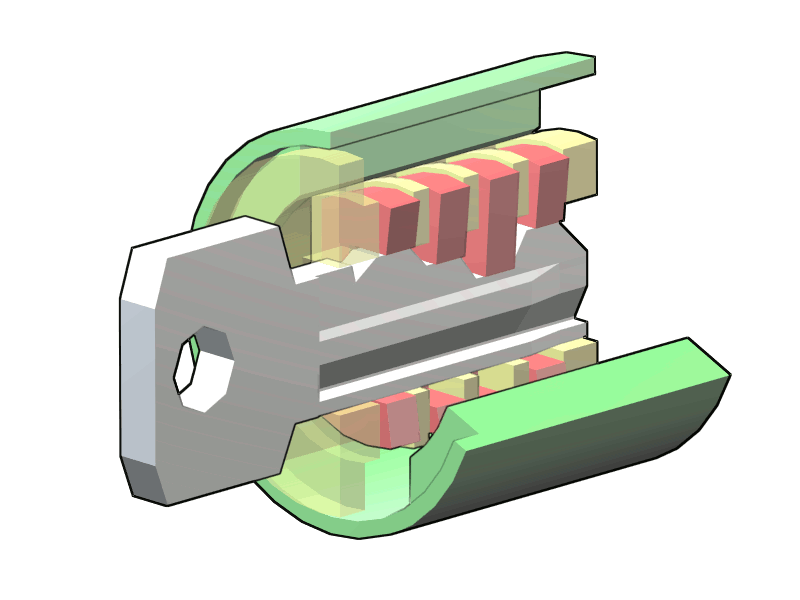
Note. By Wagaret, 2004, transferred from Wikipedia. (https://commons.wikimedia.org/wiki/File:Disc_tumbler_with_key.png) CC BY 2.0
ACh then moves across the cleft to bind to the acetylcholine receptors (AChRs) on the binding site, which in this case, is the end of the muscle fiber. The end of the muscle fiber is known as the motor end plate, which is highly folded so it can fit more AChRs on it. Once the ACh binds, it’s able to trigger depolarization. This depolarization is enough to cause something called an endplate potential (EPP). The change in voltage as a result of the EPP is enough to push the neuron to fire an action potential, which travels down the muscle fiber and causes the muscle to contract (2, Petrov et al., 2018).
Figure 3
Diagram representing a neuromuscular junction, showing ACh receptors and mitochondria of the neuron
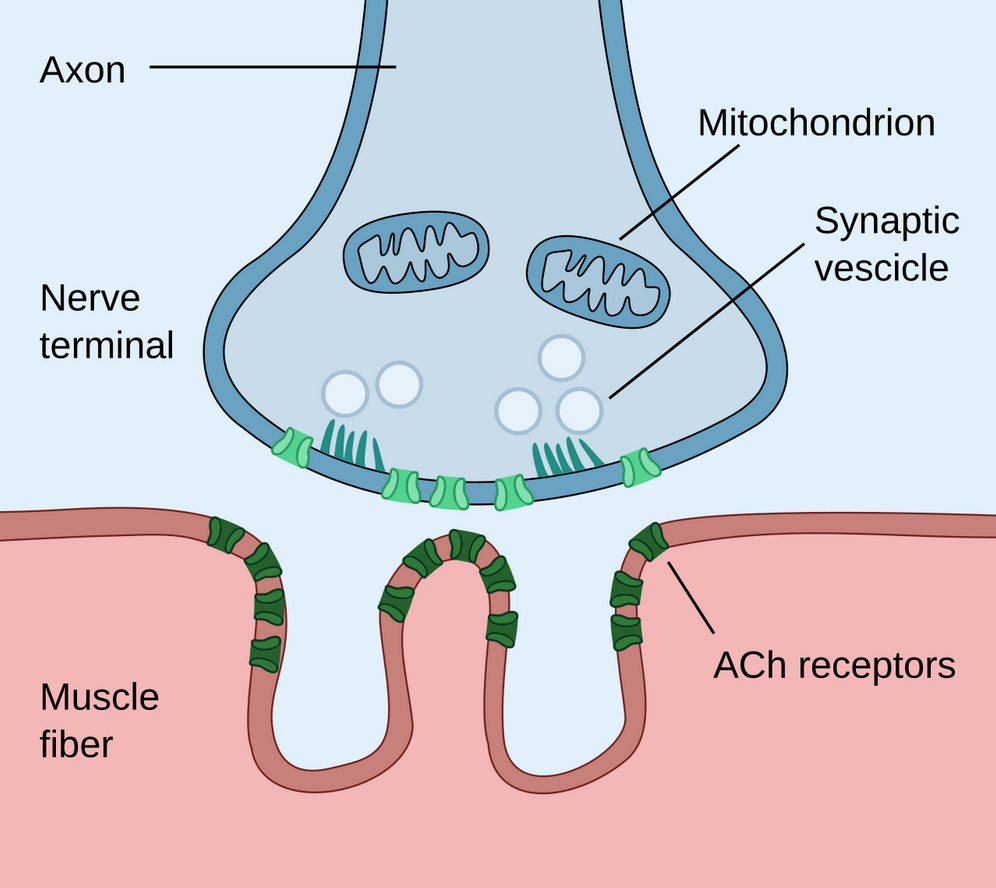
Note. By Paul Hege, 2020, own work. (https://commons.wikimedia.org/wiki/File:Neuromuscular_junction.svg) CC BY 2.0
To return to acetylcholine, what happens to it after it binds to the receptors? Basically, it needs to go away after it fulfils its use. One way it’s broken down is by degradation, which takes place by specialized enzymes, in this case, acetylcholinesterase (AChE). This has to occur to stop the muscle fiber from being continuously stimulated, which can lead to overtiring of the muscles. We can see those effects through toxins such as strychnine, where the muscles continually contract until they give up, including the heart and lungs. After AChE does its thing, the remaining products are taken back up into the neuron in order to be recycled to make more ACh. Now that we’ve covered the system upon which the neurotoxins we’ll discuss today, let’s get into the neurotoxins themselves.
Curare
Not to sound like a total nerd, but curare’s mechanism is one of the ways I first got interested in neurotoxins in the first place. One of the coolest things about this toxin is that it can be consumed with zero negative effects, but as soon as it enters through an open wound, it takes effect. The primary way to obtain curare is by using a vine, creatively named the curare vine, which is found in South America. This vine has historically been used by indigenous groups in the Orinoco River Basin in the Amazon Rainforest, as they can coat arrows or spears to hunt, first noted by Europeans around 1555. As soon as the coated weapon sinks its way into the animal, it quickly enters the bloodstream and paralyzes the prey. However, it also has uses beyond subduing prey.
Once scientists in North America observed its effects, they quickly began to incorporate the active component of curare, d-tubocurarine, into their anesthetic practice to keep patients still while they underwent surgery. The first patient was in 1942, where a 20-year-old boy needed his appendix removed. The surgery turned out to be a success, as the curare was able to keep the abdomen relaxed (3, Tyrrell et al., 1967). It’s important to note that the paralysis as a result of the curare administration would require artificial respiration to be used to keep patients breathing during the surgeries. No negative effects were recorded after curare use during surgeries, but curare’s use as a muscle relaxant eventually fell out of use since it notably didn’t dull the pain of active surgery. Now that we’ve covered it’s historical use, let’s get into how exactly it works.
As expected, curare works on ACh systems in the NMJ. Its exact mechanism of action is as a blocker, or antagonist, to the AChR sites. An antagonist is a substance that blocks the receptor site of a particular intended molecule, preventing it from binding. To bring back the lock and key analogy, you could think of an antagonist as a piece of duct tape blocking the key from being able to enter the lock. Since the active component of curare blocks the acetylcholine binding sites, acetylcholine predictably cannot bind, meaning the actions that acetylcholine usually would trigger can’t take place. Acetylcholine can’t trigger the muscle to contract, leading to paralysis. Hence why paralysis is the effect of curare administration and its use in anesthesia in historical practices.
While not used in anesthesia anymore, d-tubocurarine, the active component of curare, still has its place in modern-day research. In one such case, d-tubocurarine served as a model to test potential cures for a motor disorder called myasthenia gravis (4, Ball and Westhorpe, 2006). Myasthenia gravis is characterized by muscle weakness, fatigue, droopy eyelids, blurred or double vision, limited facial expressions, and struggling with movements such as walking or speaking. This disorder results from one’s immune system attacking itself, which leads to the destruction of AChR sites. Since the sites are destroyed, ACh cannot bind and take action, similar to what we just discussed about curare acting as an antagonist for those receptors. This eventually led to the use of another neurotoxin, physostigmine, being used to treat myasthenia gravis, as the research proved it acted as a antagonist to the effects of curare.
Physostimine is interesting within itself, as it’s the chemical compound of the Calabar bean found in western Africa. Its primary use by indigenous people was as an “ordeal poison” to serve as the judge of whether or not someone was innocent. The trick to being declared “innocent” was to swallow it quickly and induce vomiting, meaning you survived. The alternative was, well, dying and being seen as guilty by that virtue. Using physostigmine in curare poisoning models provided temporary relief and improvements in motor movements, leading to the further developments in treatment of myasthenia gravis (5, Ball and Westhorpe, 2006).
In contemporary settings, curare has been used to block the response of acetylcholine receptors to as part of treatment of nerve agent poisoning. Nerve agent poisonings primarily occur by the inhibition of acetylcholinesterase. This is bad, as it leads to the buildup of ACh in the muscle cells, leading to their overstimulation to the point of collapse (6, Whitmore et al., 2020). I provided the example of strychnine during the overview of ACh systems as having the same symptoms, but two examples of nerve agents that have been used in chemical warfare include Sarin and Novichok. Sarin was first developed by the Nazis as a colorless nerve agent; however it was later stockpiled by the U.S., USSR, and NATO. While never used in active warfare, it has been used in ethnic cleansing of the Kurd population in Iraq by Saddam Hussein and later used in Syria against rebel groups. In the case of Novichok, it was first developed in the Soviet Union, similar to Sarin, as a colorless and odorless nerve agent. Later discovered by European countries, the toxin came to light before it could be used in widescale warfare. However, it was used against a small number of Russian dissenters (7, Hubbard, 2023). These cases only highlight the importance of the development of treatment of these nerve agents, and curare plays a role in that. While it has been used in the treatment of myasthenia gravis and nerve agent poisonings, these have also required the efforts of another toxin we’ll discuss: atropine.
Figure 4
Biological Effects of Sarin in the Neuromuscular Junction
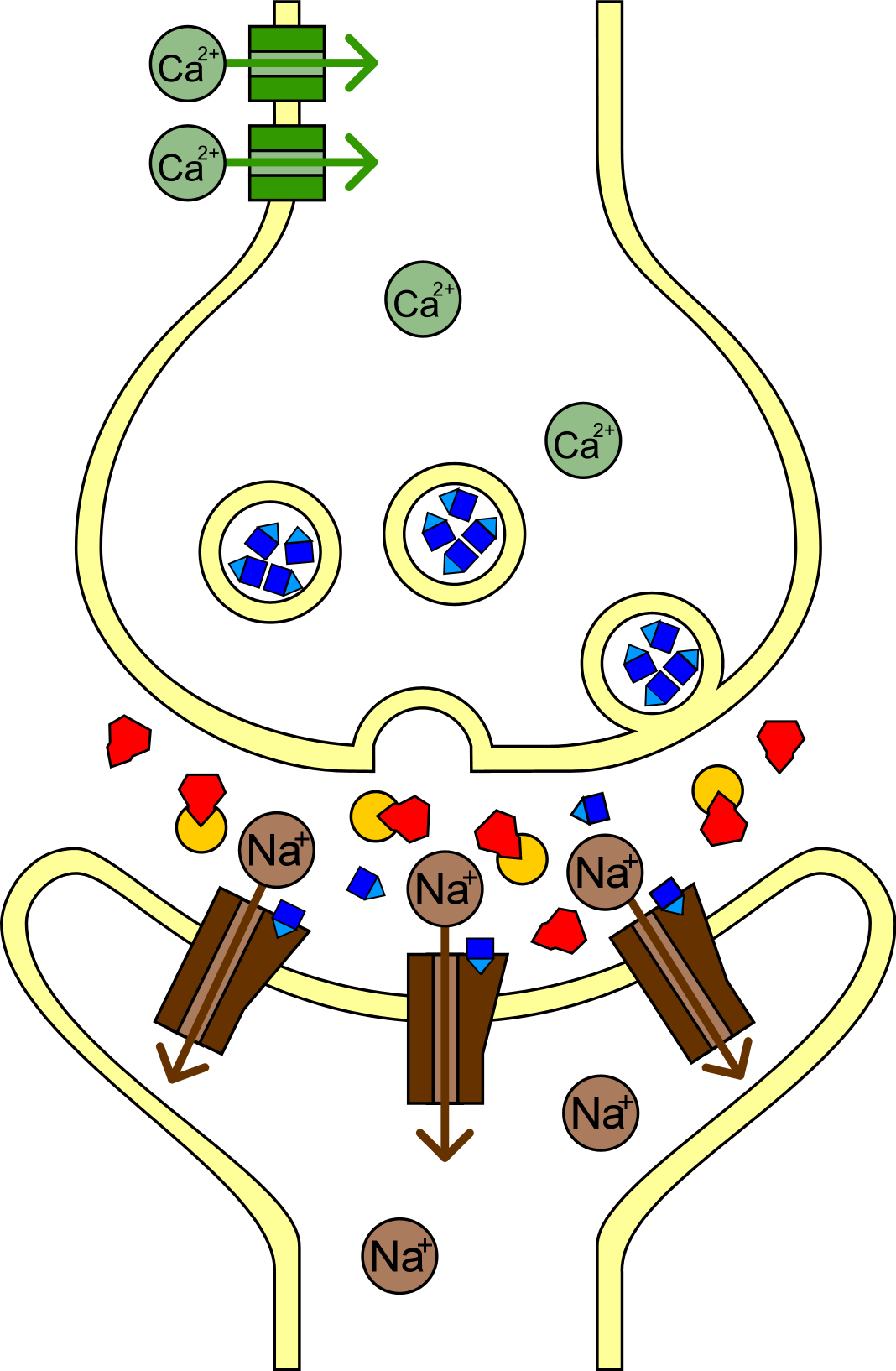
Note. Valentin Muller, 2005, own work. https://commons.wikimedia.org/wiki/File:Sarin_Wirkungsweise.png CC BY 2.0
Atropine
If you’re familiar with the works of Agatha Christie, you may have heard of atropine, a poison chosen in fiction for its ability to dissolve in liquids and odorless characteristics. Potatoes, eggplants, tomatoes, and more all come from the family Solanceae, or as it’s more commonly known, nightshades. The most infamous of these is Atropa Belladonna, or deadly nightshade. It gets its name from Atropos, the figure in Greek myth who cuts the thread of life. Belladonna comes from Italian for “beautiful woman”, where upper-class women would put drops of distilled deadly nightshade in their eyes to give them a doe-eyed appearance.
Similar to curare and tubocuranine, atropine acts a blocker to the AChRs leading to a number of symptoms. The mouth becomes “dry as a bone”, the eyes to relax focus to the point of being “blind as a bat”, blood vessels in the skin open, causing the victim to appear “red as a beet”, temperature dysregulates (“hot as a hare”), and finally hallucinations, incoherent speech, and stumbling steps (“mad as a hatter”) (8, Bradbury, 2022).
As previously mentioned, atropine is one of the most common approaches to treating nerve agent poisoning. This is done by blocking the binding of ACh to its receptors, just like with curare. By blocking these channels, it helps the systems flush excess ACh out faster. Acting as an anti-convulsant, the primary function is to avoid seizures in patients experiencing nerve agent poisoning so their bodies can return to normal levels of acetylcholine (6, Whitmore et al., 2020).
Coincidentally, atropine also has its uses when combined with physostigmine. My anecdote about its use in Italy did, in fact, have a point. The administration of physostigmine in the treatment of a number of eye issues such as damage to the nerves of the eye leading to blindness, or glaucoma, was beginning to be established in the 1800s. However, researchers found that the physostigmine constricted the pupil to extreme levels, the reaction normally observed in dim lighting. With the addition of atropine, which dilates the pupil, the drawbacks were able to be combated (9, Katz and Barohn, 2021). With that, we move onto our final neurotoxin: botulinum toxins.
Botulinum Toxins
Figure 5
Botulism Mechanism
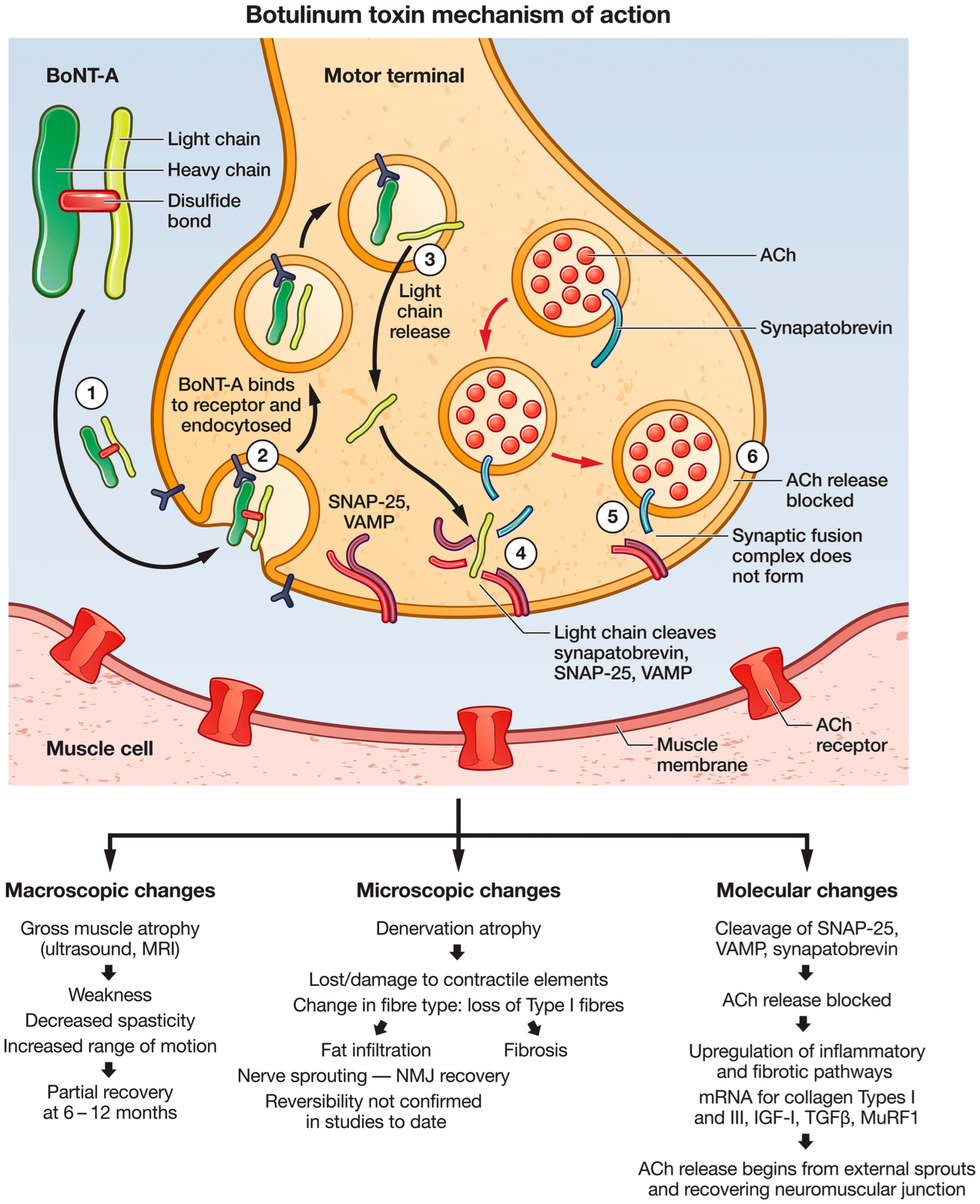
Note. Parisa Gazerani, 2022, own work.
https://commons.wikimedia.org/wiki/File:Botulism.png CC BY 2.0
Most people would be most familiar with the Botulinum group of toxins for its use in cosmetic procedures under the name Botox. Overall, the produced effect of these toxins is paralysis. While there is no treatment or cure for the excess of botulinum toxins, or botulism, the amounts administered during treatments are perfectly safe. In the case of Botox, a certain area of muscles is targeted. In the treatment of motor disorders such as dystonia, acetylcholine systems at large are targeted throughout the body. The paralysis is produced by removing some of the proteins that help move the vesicles containing ACh to the end of the synapse. Since the proteins are removed, the vesicle can’t reach the end and release the acetylcholine that it contains (10, Yoelin and Hooper, 2024). As the ACh can’t be released, none of the effects it normally allows to take place can occur, leading to no muscle contraction.
While the vast majority of treatments involving botulinum toxins are cosmetic, such as wrinkles and scars, other avenues are being pursued. Botulinum treatments have been introduced in areas such as migraine treatments and excessive twitching, but its use in motor disorders has been a new area of research. The most common motor disorder that has been pursued under these treatments has been dystonia. Dystonia is accompanied by symptoms of involuntary and repetitive muscle contractions. These movements are often painful for those experiencing them. However, with the use of botulinum toxins, these symptoms can be alleviated. Studies have found that these toxins can reduce symptoms for more than 12 years, which is a huge relief to those living with dystonia. At the start of treatment, patients reported some muscle weakness, but these effects quickly disappeared. The botulinum treatments were found not only to treat the motor symptoms, but also to relieve pain (11, Albanese et al., 2015). Overall, these toxins can be used to treat the suffering, not just cause it.
References
Omar, A., Marwaha, K., & Bollu, P. C. (2023, May 1). Physiology, Neuromuscular Junction. Nih.gov; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK470413/
Petrov, K. A., Nikolsky, E. E., & Masson, P. (2018). Autoregulation of Acetylcholine Release and Micro-Pharmacodynamic Mechanisms at Neuromuscular Junction: Selective Acetylcholinesterase Inhibitors for Therapy of Myasthenic Syndromes. Frontiers in Pharmacology, 9. https://doi.org/10.3389/fphar.2018.00766
Tyrrell, M. F., Scurr, C. F., & Davison, M. H. (1967). The curare exhibition. Marking the 25th anniversary of the introduction of the drug into clinical anaesthesia. Annals of the Royal College of Surgeons of England, 41(5), 424. https://pmc.ncbi.nlm.nih.gov/articles/PMC2312021/
Ball, C., & Westhorpe, R. (2005). Muscle Relaxants—d-tubocurarine. Anaesthesia and Intensive Care, 33(4), 431–431. https://doi.org/10.1177/0310057×0503300401
Ball, C., & Westhorpe, R. N. (2006, August). Muscle Relaxants- Reversal Agents. Sage Journals; Australian Society of Anaesthetists. https://doi.org/10.1177/0310057X0603400420
Whitmore, C., Lindsay, C. D., Bird, M., Gore, S. J., Rice, H., Williams, R. L., Timperley, C. M., & Green, A. C. (2020). Assessment of false transmitters as treatments for nerve agent poisoning. Toxicology Letters, 321, 21–31. https://doi.org/10.1016/j.toxlet.2019.12.010
Hubbard, B. (2023). Poison: The History of Potions, Powders and Murderous Practitioners.
Bradbury, N., PhD. (2022). A taste for poison: Eleven Deadly Molecules and the Killers Who Used Them. St. Martin’s Press.
Katz, N. K., & Barohn, R. J. (2020). The history of acetylcholinesterase inhibitors in the treatment of myasthenia gravis. Neuropharmacology, 182, 108303. https://doi.org/10.1016/j.neuropharm.2020.108303
Yoelin, S., & Hooper, D. (2024). New and Future Developments in Neurotoxins. Dermatologic Surgery, 50(9S), S112–S116. https://doi.org/10.1097/dss.0000000000004346
Albanese, A., Romito, L. M., & Calandrella, D. (2015). Therapeutic advances in dystonia. Movement Disorders, 30(11), 1547–1556. https://doi.org/10.1002/mds.26384
Kuznetstov, A. (2007). Bottle of poison. File:Poison.jpg. Retrieved 2026, from https://commons.wikimedia.org/wiki/File:Poison.jpg.
Hege, P. (2020). Diagram representing a neuromuscular junction, showing ACh receptors and mitochondria of the neuron. File:Neuromuscular junction.svg. Retrieved 2026, from https://commons.wikimedia.org/wiki/File:Neuromuscular_junction.svg.
Wapcaplet. (2004). Illustration of the en:disc tumbler lock created by Wapcaplet in Blender and touched up in the en:GIMP. en:Category:Images Created with Blender. File: Disc Tumbler with key.png. Retrieved 2026, from https://commons.wikimedia.org/wiki/File:Disc_tumbler_with_key.png.
Muller, Valentin (2005). Biological Effects of Sarin in the Neuromuscular Junction. File:Sarin Wirkungsweise.png. Wikimedia Commons, November 16. Retrieved 2026, from https://commons.wikimedia.org/wiki/File:Sarin_Wirkungsweise.png
Gazerani, P. (2022). Botulism Mechanism. File:Botulism.png. Retrieved 2026, from https://commons.wikimedia.org/wiki/File:Botulism.png.
