Menopause is the moment in a person’s life where menstruation has permanently ceased, and is accompanied by many intense physiological changes. Despite the fact that over half of the global population will experience menopause, it remains critically underfunded and understudied. The National Institute of Health (NIH), which is the biggest public funder of behavioral and biomedical research in the world, did not have a specific funding category for “Menopause” until 2023 (1). In 2024, less than a quarter-of-a-percent of NIH money dedicated to Research Project Grants was spent on menopause-focused research (1, 2). This exemplifies a broader societal trend and consequently, it is unsurprising that most people have not received adequate education about menopause, even those who will one day undergo menopause themselves.
Formal education and readily accessible, accurate information regarding menopause-related symptoms, biology, and even what menopause is, is scarce (3-5). A U.K. based study aiming to assess the prevalence of menopause-related education gathered 947 participants, 97% of whom were women. When prompted about menopause-related education, more than 90% of participants said they had not received any formal education about menopause in school before college (4). These numbers are incredibly disappointing and highlight the urgent need for expanded educational opportunities about menopause.
For those who experience menopause or care for someone experiencing menopause, knowledge is power. Educating women about menopause has been demonstrated to improve their quality of life and, in many cases, mitigate menopause-related symptoms (3). Becoming more educated about menopause will allow us to better support our future selves and/or loved ones who undergo menopause.
What is Menopause? Differentiating Perimenopause, Menopause, and Postmenopause
There are several definitions related to menopause that are misunderstood or get confused with one another. One example of this is the difference between menopause and postmenopause. While menopause specifically refers to the point in time where menstruation has permanently ceased after being absent for 12 consecutive months, postmenopause refers to any period of life after menopause has been diagnosed (6). Perimenopause is the period of time leading up to menopause and usually lasts around 4 years, but can vary in length depending on the person (5). During perimenopause, irregular menstrual cycles and intense hormonal fluctuations occur (7). Hormones are important chemical messengers that help coordinate various functions in the body.
Hormonal fluctuations and other factors can influence the various symptoms experienced by perimenopausal women (7). Some of the most commonly reported menopause-related symptoms are muscle and/or joint pain, exhaustion, sleep disturbances, depression, anxiety, vaginal dryness, and hot flashes (8, 9). For many women, menopause-associated symptoms/experiences can bring substantial changes and challenges to day-to-day living, including at work, during social activities, or to overall quality of life (11). Additionally, menopausal symptoms can interact with other factors (such as those visualized in Figure 1) to compound and potentially worsen mental and physical health (10). Therefore, when addressing menopause-related concerns, it is important to recognize contributing factors that are both menopause-related and menopause-independent.
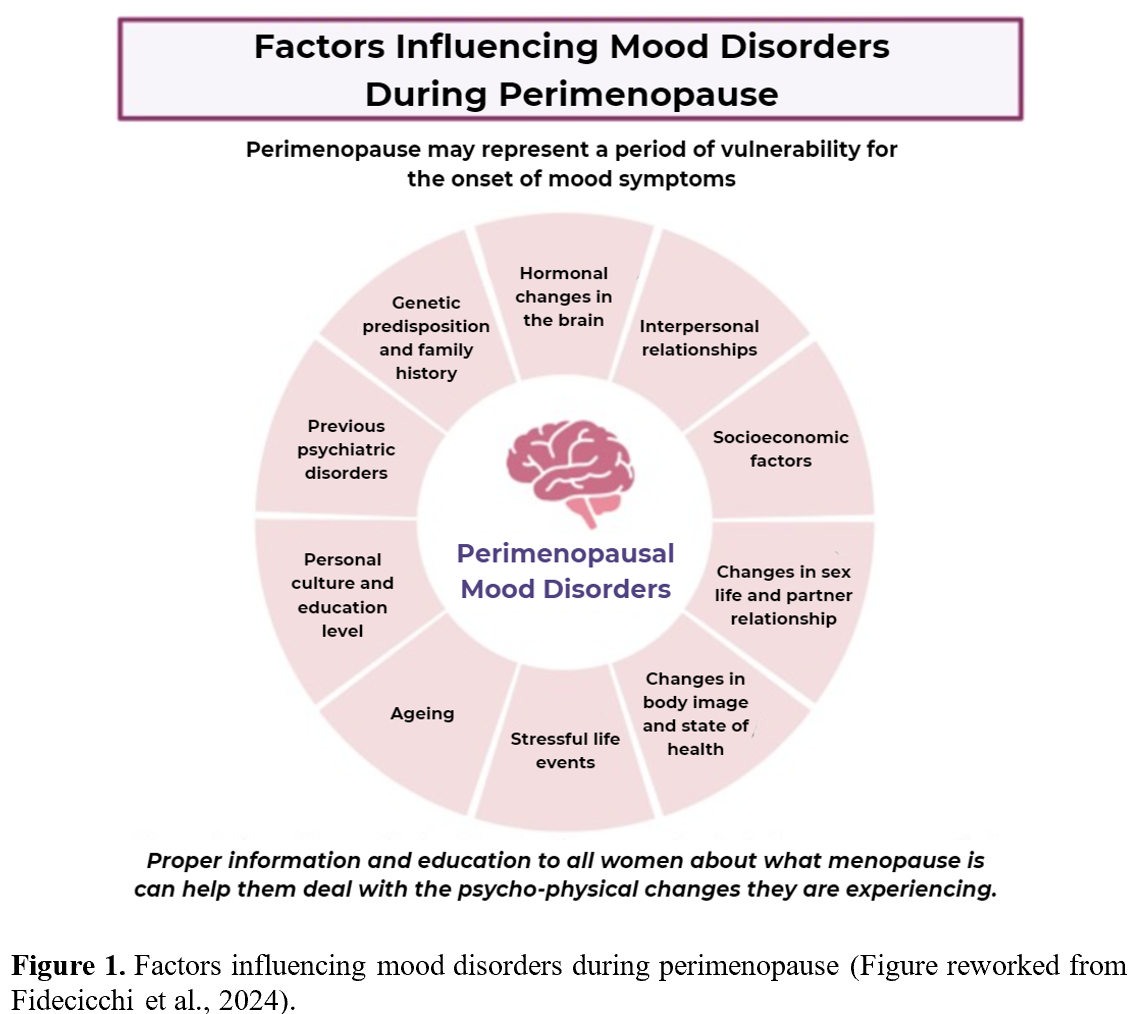
Unfortunately, menopausal symptoms and subsequent life changes do not vanish after perimenopause and oftentimes continue in postmenopausal women. In a better-case scenario, certain symptoms are the most severe during perimenopause and become increasingly manageable as time goes on. Two less-favorable scenarios are that symptoms retain similar severity or progressively worsen with age (12). Either way, dealing with menopausal symptoms can be frustrating and stressful, especially with the lack of accurate education accessible to the public.
Estrogen’s Contribution to Menopausal Symptoms
During perimenopause, acute shifts in estrogen levels occur, oftentimes leading to the onset of menopausal symptoms. Estrogen is one of the most recognized hormones to be impacted by menopause, and functions throughout life to regulate bone, heart, and brain health, as well as menstruation and reproduction (13). Perimenopausal women experience severe fluctuations and general downward trends in estrogen levels. During the year leading up to menopause, estrogen levels tend to drop the most substantially (9). Once menopause hits, estrogen levels continue to drop, although typically in a more gradual manner. Fluctuations and drops in estrogen levels throughout the body, including in the brain, have been associated with many previously mentioned symptoms like depression, exhaustion, hot flashes, etc. (8, 9). In postmenopausal women specifically, loss of estrogen likely increases vulnerability for cognitive decline and osteoporosis (14, 15). Estrogen works in complex ways to support numerous bodily functions, which estrogen loss during peri-and-post-menopause can undermine.
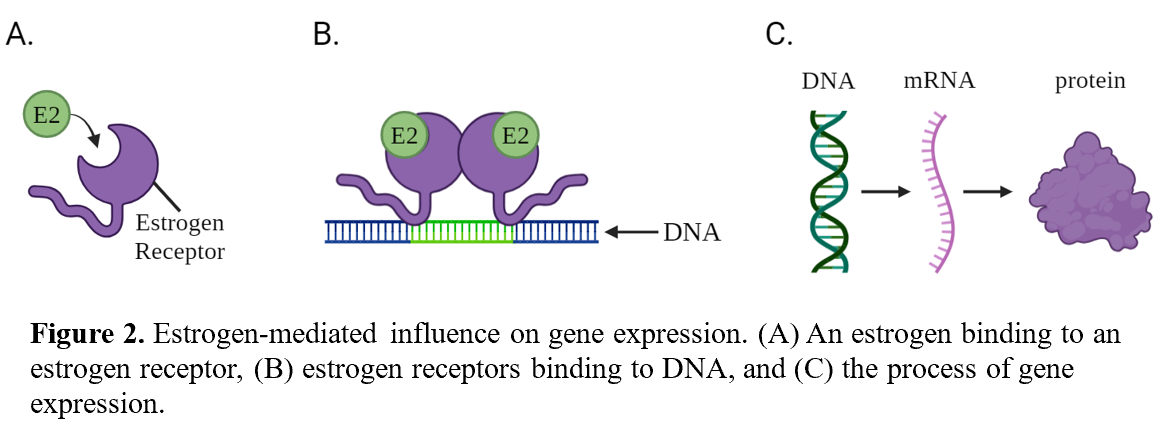
Estrogen by itself is usually not enough to produce an effect in the brain. Estrogen interacts with complex networks of other chemical messengers, proteins, and additional neural structures to influence the brain (10). To accomplish this, estrogen needs to bind to an estrogen receptor, which is a protein specialized to mediate estrogen-driven effects in the brain (13). Estrogens and estrogen receptors function like a lock and key, where estrogen receptors are the lock and estrogens are the key (Figure 2A). Once an estrogen binds to, or “unlocks”, the estrogen receptor, the “unlocked” receptor can initiate changes within the brain. For instance, bound, or “unlocked”, estrogen receptors group up with other “unlocked” estrogen receptors and bind to specific DNA sites (Figure 2B; 13).
By binding to DNA sites, estrogen receptors can help regulate one of the most powerful biological processes: gene expression (Figure 2C; 13). The process of gene expression begins with DNA, which is like a cherished recipe book that your grandma swears by (that happens to contain all of our genetic information). The DNA recipe is “read” by proteins which then construct what is called mRNA. mRNA is like a notecard on which you wrote one recipe from Grandma’s book so that you could attempt to replicate her cooking in your messy dorm kitchen. The mRNA is then “read” to produce the final product: a protein – or in your kitchen, a (hopefully) good meal.
Estrogen receptors play a role in countless processes in the brain. Notably, estrogen receptors are most prevalent in brain regions highly responsible for learning, memory, and emotional regulation (16). In some of these regions, estrogen receptors frequently promote the expression of genes associated with the production and protection of neurons (14). Furthermore, estrogen receptors also influence the brain through non-genetic pathways that do not involve DNA-binding, such as by interacting with other chemical messengers. One well-known chemical messenger that estrogen associates with is serotonin. Studies suggest that estrogen may act to maintain serotonin levels in the brain (10). This is notable because serotonin helps regulate many important bodily functions, such as mood, cognitive function, and sleep (10). As a result, when estrogen-mediated gene expression or chemical interactions are disrupted, neurological well-being can be disturbed, which contributes to some of the symptoms observed in menopause.
Understanding estrogen-mediated changes in the brain is highly relevant to understanding why certain menopausal symptoms may occur. When estrogen levels drop during perimenopause and postmenopause, less estrogen is available to bind to estrogen receptors. Consequently, estrogen’s support of important physiological processes is undermined (13, 10). Considering that estrogen supports the production and protection of neurons and the activity of serotonin in the brain, decreased estrogen levels can leave the brain vulnerable to instability (10, 14, 17). Such instability likely contributes to peri-and post-menopausal women being more likely to acquire certain types of mental health disorders and cognitive decline (14, 15). Learning about the neurological changes occurring during peri-and post-menopause can help identify and mitigate symptoms, as well as help in finding potential medical treatments for such symptoms.
How Do We Look at Estrogen Receptors in the Brain?
Many scientific tools can be used to study estrogen’s role in menopause, one of which is the PET scan. PET scans are often administered to detect disease occurrence, drug activity, or the presence of a biological component in the body. PET scans are completed at hospitals or specialized research facilities that operate the large, donut-shaped PET scanner. Before a PET scan, patients are either given a pill or are injected with a solution containing a “tag” designed to detect a particular substance. The PET scanner can pick up signals from this “tag” (18).
The presence of estrogen receptors in the brain has been examined through PET scan usage. One study investigated how estrogen receptor presence in the brain changes throughout pre-, peri-, and post-menopausal stages of life (15). The results of this study indicate that postmenopausal brains contain more unbound estrogen receptors on average than premenopausal brains (15). This may seem counterintuitive, but the “tag” used within this study detects estrogen receptors by binding to them, meaning that detected estrogen receptors have not been bound by estrogens. Therefore, increased estrogen receptor detection signifies lower estrogen binding, indicating decreased estrogen-mediated effects on the brain. Thankfully, as both scientists and the public learn more about how the brain is influenced by menopause, it is possible that new and/or improved treatments will be made available.
Mitigating Menopausal Symptoms Using Hormone Therapy – Can We Trust It?
We know how to look at estrogen receptors in the brain. We also know that estrogen levels drop leading up to menopause… so what can we do about it? One highly effective treatment – yet highly controversial – is hormone therapy. Estrogen-based hormone therapies are medications containing small doses of estrogen, sometimes in combination with another hormone (19). These medications come in many forms, like gels, pills, patches, etc.
There are many controversies surrounding estrogen-based treatments for menopause. From 2003-2025, the Food and Drug Administration (FDA) instituted “black box” warnings for estrogen-based medications approved to treat menopausal symptoms and osteoporosis (19). A black box warning is the most severe safety label the FDA can place onto medications and signifies that the medication may increase one’s likelihood for developing substantial, and sometimes life-threatening, health risks. The FDA made this decision based on a 2002 Women’s Health Initiative (WHI) study that examined health outcomes for an estrogen-based hormone therapy in older, postmenopausal women. The WHI study communicated an increased risk for cardiovascular events and breast cancer in women taking the estrogen-based treatment compared to women not taking the treatment (19-21). The placement of black box warnings on menopause-related estrogen therapies lead to doctors prescribing the medications at much lower rates and women avoiding these medications out of fear for developing breast cancer, cardiovascular issues, or any other warning associated with the label (19, 21).
Nonetheless, many experts in women’s heath have long considered estrogen hormone therapies to be the gold standard for menopause-related care. This brings up a question: why were women steered away from receiving these treatments for such a long time? Certain concerns arose regarding the original WHI study used by the FDA to justify the addition of black box labels to estrogen-based menopausal therapies. The study only included postmenopausal women, the vast majority of which were above the age of 60 or had been postmenopausal for over 10 years (20). In more recent research, evidence suggests that it can be crucial to begin estrogen-based menopausal therapies within 10 years of menopause onset and below the age of 60. Starting these therapies at later points is associated with mildly increased risks for certain negative health outcomes like cardiovascular events, stroke, and breast cancer (22, 23). If administered at earlier timepoints, these risks largely disappear (22). Some studies have suggested that appropriately administered estrogen therapy can reduce incidences of cardiovascular-related fatalities, mitigate cognitive decline, provide general relief from menopausal symptoms, and even offer mild protection from Alzheimer’s disease (19, 22), although data is limited in this regard.
In late 2025, the FDA removed black box warnings from estrogen-based medications approved to treat menopausal symptoms. This decision resulted from years of public comments, surmounting scientific evidence, and expert panels suggesting that the FDA should revisit the 2002 evaluation of menopausal hormone therapies (19). Although the 2002 WHI study appears to have overemphasized the dangers of estrogen-based treatments in postmenopausal women, it still provided a framework enabling future researchers to realize the importance of early treatment and the risks of long-term hormone use. Nonetheless, the FDA’s decision to apply generalized and overstated warnings from this WHI study was costly, as many women lost out on potentially life-changing treatments. While the removal of the black box label does not indicate that no risks are involved with estrogen therapies, it emphasizes the need for tailored treatment recommendations, not sweeping generalizations (19).
Future developments in scientific research and education regarding menopause are necessary for women to make informed decisions about their health. Many women have not been provided adequate education about menopause, which undermines their ability to identify and manage symptoms, seek appropriate treatment, and support themselves and each other. Furthermore, controversies surrounding menopause therapy options make symptom management all the more confusing. Although national funding for menopause-centered research is largely out of the general population’s control, pushing for menopause education can still make a big difference. Remember, knowledge is power.
References
- NIH Data Book—NIH Budget History. (n.d.). Retrieved April 22, 2026, from https://report.nih.gov/nihdatabook/category/1
- RePORT. (n.d.). Retrieved April 22, 2026, from https://report.nih.gov/funding/categorical-spending#/
- McFeeters, C., Pedlow, K., McGinn, D., & McConnell, K. (2024). A rapid review of menopausal education programmes. Archives of Women’s Mental Health, 27(6), 975–983. https://doi.org/10.1007/s00737-024-01476-8
- Harper, J. C., Phillips, S., Biswakarma, R., Yasmin, E., Saridogan, E., Radhakrishnan, S., C Davies, M., & Talaulikar, V. (2022). An online survey of perimenopausal women to determine their attitudes and knowledge of the menopause. Women’s Health, 18, 17455057221106890. https://doi.org/10.1177/17455057221106890
- Wegrzynowicz, A. K., Walls, A. C., Godfrey, M., & Beckley, A. (2025). Insights into Perimenopause: A Survey of Perceptions, Opinions on Treatment, and Potential Approaches. Women (Basel, Switzerland), 5(1), 4. https://doi.org/10.3390/women5010004
- Yang, J. L., Hodara, E., Sriprasert, I., Shoupe, D., & Stanczyk, F. Z. (2024). Estrogen deficiency in the menopause and the role of hormone therapy: Integrating the findings of basic science research with clinical trials. Menopause, 31(10), 926. https://doi.org/10.1097/GME.0000000000002407
- Delamater, L., & Santoro, N. (2018). Management of the Perimenopause. Clinical Obstetrics and Gynecology, 61(3), 419–432. https://doi.org/10.1097/GRF.0000000000000389
- Joseph, N., Nagaraj, K., Saralaya, V., Nelliyanil, M., & Rao, P. J. (2014). Assessment of menopausal symptoms among women attending various outreach clinics in South Canara District of India. Journal of Mid-Life Health, 5(2), 84–90. https://doi.org/10.4103/0976-7800.133996
- Santoro, N. (2016). Perimenopause: From Research to Practice. Journal of Women’s Health, 25(4), 332–339. https://doi.org/10.1089/jwh.2015.5556
- Fidecicchi, T., Giannini, A., Chedraui, P., Luisi, S., Battipaglia, C., Genazzani, A. R., Genazzani, A. D., & Simoncini, T. (2024). Neuroendocrine mechanisms of mood disorders during menopause transition: A narrative review and future perspectives. Maturitas, 188, 108087. https://doi.org/10.1016/j.maturitas.2024.108087
- Arar, M. A., & Erbil, N. (2023). The effect of menopausal symptoms on women’s daily life activities. Przegla̜d Menopauzalny = Menopause Review, 22(1), 6–15. https://doi.org/10.5114/pm.2023.126436
- Ahsan, M., Mallick, A. K., Singh, R., & Prasad, R. R. (2015). Assessment of menopausal symptoms during perimenopause and postmenopause in tertiary care hospital. Journal of Basic and Clinical Reproductive Sciences, 4(1), 14-19. https://doi.org/10.4103/2278-960X.153516
- Fuentes, N., & Silveyra, P. (2019). Estrogen receptor signaling mechanisms. Advances in Protein Chemistry and Structural Biology, 116, 135–170. https://doi.org/10.1016/bs.apcsb.2019.01.001
- Cho, J. M., Lee, J., Ahn, E.-M., & Bae, J. (2025). Beyond Hot Flashes: The Role of Estrogen Receptors in Menopausal Mental Health and Cognitive Decline. Brain Sciences, 15(9), 1003. https://doi.org/10.3390/brainsci15091003
- Mosconi, L., Nerattini, M., Matthews, D. C., Jett, S., Andy, C., Williams, S., Yepez, C. B., Zarate, C., Carlton, C., Fauci, F., Ajila, T., Pahlajani, S., Andrews, R., Pupi, A., Ballon, D., Kelly, J., Osborne, J. R., Nehmeh, S., Fink, M., … Brinton, R. D. (2024). In vivo brain estrogen receptor density by neuroendocrine aging and relationships with cognition and symptomatology. Scientific Reports, 14(1), 12680. https://doi.org/10.1038/s41598-024-62820-7
- Almey, A., Milner, T. A., & Brake, W. G. (2015). Estrogen receptors in the central nervous system and their implication for dopamine-dependent cognition in females. Hormones and Behavior, 74, 125–138. https://doi.org/10.1016/j.yhbeh.2015.06.010
- Hara, Y., Waters, E. M., McEwen, B. S., & Morrison, J. H. (2015). Estrogen Effects on Cognitive and Synaptic Health Over the Lifecourse. Physiological Reviews, 95(3), 785–807. https://doi.org/10.1152/physrev.00036.2014
- What Is a PET Scan. (2026). Cleveland Clinic. Retrieved May 4, 2026, from https://my.clevelandclinic.org/health/diagnostics/10123-pet-scan
- Makary, M. A., Nguyen, C. P., Høeg, T. B., & Tidmarsh, G. F. (2026). Updated Labeling for Menopausal Hormone Therapy. JAMA, 335(2), 117–118. https://doi.org/10.1001/jama.2025.22259
- Rossouw, J. E., Anderson, G. L., Prentice, R. L., LaCroix, A. Z., Kooperberg, C., Stefanick, M. L., Jackson, R. D., Beresford, S. A., Howard, B. V., Johnson, K. C., Kotchen, J. M., & Ockene, J. (2002). Risks and Benefits of Estrogen Plus Progestin in Healthy Postmenopausal Women: Principal Results From the Women’s Health Initiative Randomized Controlled Trial. JAMA: The Journal of the American Medical Association, 288(3), 321–333. https://doi.org/10.1001/jama.288.3.321
- Zeeshan, F., & Saqlain, A. (2026). FDA’s 2025 removal of black box warnings on menopausal hormone therapy. Annals of Medicine and Surgery, 88(2), 2222–2223. https://doi.org/10.1097/MS9.0000000000004749
- Kim, J.-E., Chang, J.-H., Jeong, M.-J., Choi, J., Park, J., Baek, C., Shin, A., Park, S. M., Kang, D., & Choi, J.-Y. (2020). A systematic review and meta-analysis of effects of menopausal hormone therapy on cardiovascular diseases. Scientific Reports, 10(1), 20631. https://doi.org/10.1038/s41598-020-77534-9
- Manson, J. E., Chlebowski, R. T., Stefanick, M. L., Aragaki, A. K., Rossouw, J. E., Prentice, R. L., Anderson, G., Howard, B. V., Thomson, C. A., LaCroix, A. Z., Wactawski-Wende, J., Jackson, R. D., Limacher, M., Margolis, K. L., Wassertheil-Smoller, S., Beresford, S. A., Cauley, J. A., Eaton, C. B., Gass, M., … Wallace, R. B. (2013). Menopausal Hormone Therapy and Health Outcomes During the Intervention and Extended Poststopping Phases of the Women’s Health Initiative Randomized Trials. JAMA, 310(13), 1353–1368. https://doi.org/10.1001/jama.2013.278040
